Incidence of melanoma has been increasing in recent years, which may be due to environmental influences and ozone layer depletion. Despite intensive research, a considerable part of melanoma can still not be treated.
Dr. Beate Rinner is trying to find new treatment strategies for rare types of NRAS-mutated melanoma. She works as the Head of the Core Facility (CF) Alternative Biomodels and Preclinical Imaging at Graz Medical University, Austria. The CF is responsible for the CellBank Graz, which offers a wide range of services and support related to cell and tissue culture to all researchers at the Medical University. The cell bank acts as a distributor and a secure repository for cell lines developed by local researchers. The aim is to make it easier and cheaper for local scientists to access highly quality-controlled cell lines.
“Our CF provides cell lines, mycoplasma and STR analyses, isolation of non-tumorigenic cells (e.g. fibroblasts, melanocytes, and keratinocytes) as control cell systems, establishment of 3D and dynamic cell culture systems, and viability and proliferation assays. One main part is the establishment and characterisation of tumor cell lines, focusing on sarcoma, chordoma, melanoma and brain tumors. Furthermore, our CF is responsible for preclinical studies like micro-CT, ultrasonic and optical imaging, and irradiation, to close the gap between in vitro and in vivo studies”, Dr. Rinner says.
The work of the Core Facility is based on the principles of 3R – replacement, refinement, and reduction. These are guiding principles for more ethical use of animals in testing.
“We have an excellent cooperation with surgeons, pathologists, dermatologists and many other specialists in University Hospital Graz. The near-localization, the personal contact, the excellent diagnostics and the consistent patient history make it possible to create high quality cell lines,” Dr. Rinner says.
Studying rare tumor cell lines
Dr. Rinner’s research team aims to find new treatment strategies by establishing and characterizing rare tumor cell lines and generating cell systems that mimic the in vivo system.
“There exist various cell lines, but they are quite old, mutated and often far from their original genotype or phenotype. And very often, the patient history, an extremely important issue, is missing. Our aim is to create cell lines with continuous patient history and to mimic the in vivo state as closely as possible. That can only be achieved with suitable 3D culture models to each tumor entity”, Rinner explains.
In their recent study, Rinner’s team have been looking at NRAS-mutant melanoma, a distinct type of melanoma that comprises 15—20 per cent of all melanomas and appears to confer a poor prognosis.
There are some specific challenges in developing melanoma cell lines and cell culture models that retain characteristics of the in vivo state.
“NRAS-mutated melanoma patients are rarer than BRAF-mutated patients. In addition to this, most cell models lose melanoma-specific markers, like melanin, in in vitro culturing.
Excellent results with GrowDex
Dr. Rinner was introduced to GrowDex during a 3-month stay abroad in Helsinki, joining the group of Dr. Marjo Yliperttula who had been working for many years with GrowDex. Dr. Rinner had been looking for a 3D cell culture matrix that was non-toxic, easy-to-handle and contained no animal products. “We decided to culture our NRAS-mutated melanoma cell line MUG-Mel2 in GrowDex as well to see the growth behavior of the cells in 3D.”
“Initially we found working with GrowDex challenging as the stock concentration is quite viscous and can be difficult to pipette. However, with perseverance and guidance from experienced GrowDex user Johanna Niklander, we overcame this and the natural growth of the cells more than made up for it.”
Dr. Rinner and research assistant Johanna Niklander tested the 3D growth ability of MUG-Mel2 to see if cells could form spheroids and if the intense stable pigmentation of cells could be reflected.
The ability of MUG-Mel2 to form spheroids could indeed be achieved by cultivation in NFC scaffolds, which was tested by using three different concentrations of NFC and two cell concentrations. GrowDex presented an optimal matrix for the cells to maintain their natural behavior and to increase the deep brown staining.
“I would recommend GrowDex for this application,” Dr. Rinner says.
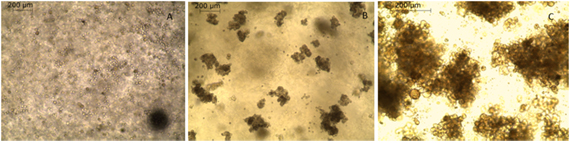
Figure 1. MUG Mel2 cell line cultured in GrowDex® hydrogel
A) Seeding of MUG MEl2 cells, formation of spheroids after one day
B) Morphology of spheroids after three days
C) Morphology of spheroids after five day, prominent brown melanin staning
Due to its phenotypically and genotypically precise characterization, the newly established cell line MUG-Mel2 presented aggressive and fast-growing behavior in mouse and chick models. With the high pigmentation, the cell line turned out to be a promising new model for innovative NRAS therapy options.
“Unfortunately, it is very unlikely that scientists could fully replace animal models, but with 3D cell culture we can for sure reduce to a minimum”, Dr. Rinner says.
The next project of Dr. Rinner and assistant Niklander is to culture primary melanoma tumor pieces directly in GrowDex to keep the whole types of melanoma cells in culture.
Image text: Dr. Beate Rinner’s research team in Graz. From the left: Marie-Therese Frisch, Alexandrea Lipfert, Katharina Meditz, Ines Anders, Alexander Stallinger and Beate Rinner (in the front). Johanna Niklander is missing from the photo.
Read the scientific report on the study
