Mechanisms for transporting living biological cells are limited and, at the very least, inefficient, restricting how much cell populations can be shared between researchers, and holding back applications where this would be especially beneficial. A chance encounter at a conference in Switzerland led to a collaboration between UPM, Cellbox Solutions GmbH and the Max Planck Institute in Münster, Germany, aiming to develop a way to successfully transport complex live cell assays.
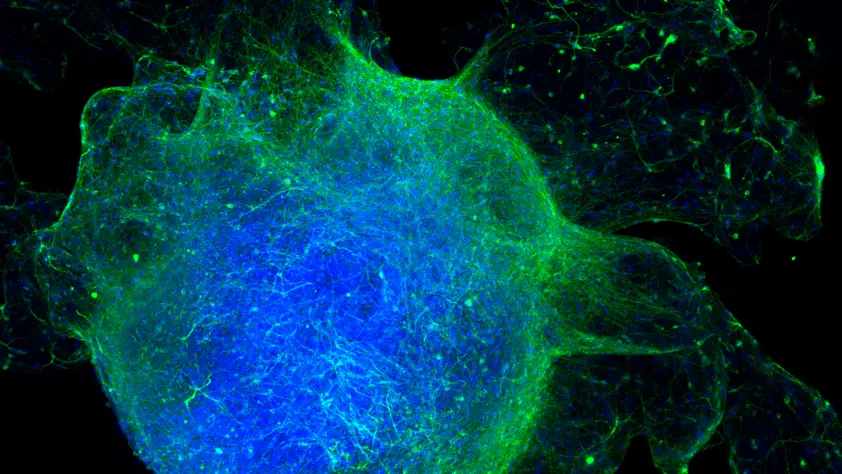
Dr Jan Bruder and his team in the department of Cell and Developmental Biology at the Max Planck Institute for Molecular Biomedicine (MPI) in Münster focus on stem cell research, specifically on high-throughput disease modeling. Since 2011, he has been developing new screening protocols for human neurodegenerative diseases, turning stem cells into the same types of neurons that die in the brains of people with diseases like Parkinson's and Alzheimer’s, and producing cellular models in a dish that more closely resemble structures in vivo. This work has led to the development of automated solutions, scaling up from manual protocols to producing tens of thousands of different samples, firstly using 2D cell culture and then, more recently, 3D alternatives that ultimately produced midbrain organoids.
In the course of these developments, Jan has trialed several different extracellular matrices (ECM), with varying success: “There are several different options on the market, but many have significant disadvantages, and this is complicated even further when automation comes into play. Even products that are considered ‘gold standard’ tend to be temperature-sensitive, sticky, and really difficult to handle on automated set-ups. With their varying composition, they often introduce variations into the cell models that we aim to standardize for industrial use, typically promoting extremely fast but unnatural growth. While in some respects this is an advantage, we found that ultrafast growth simply did not reflect the physiological process, and so looked for a more neutral ECM substrate. We discovered GrowDex®, assessing the viability of cells, as well as the expression of different markers and similar characteristics, and found essentially no differences from normal cell growth. It turned out to be impressively neutral, which is really quite remarkable for cellular support systems.”
Jan had been using GrowDex for three years when a meeting with UPM at a conference in Switzerland began a conversation about the development of a viable cell transport system. Jan explained: “The midbrain organoids we have now cultivated would be of great benefit to fellow researchers for drug discovery applications around the world, and we are considering commercialization of our 3D cell culture. In order to do that, we need to be able to ship the organoids. However,This has been a significant stumbling block because we can see an exponential fall in cell activity within 20 minutes of taking them out of an incubator, so this pilot project was a great idea.”
Tony Kiuru, Business Development Manager at UPM, continued: “We knew that Jan was interested in transporting cells and we had some knowledge of the Cellbox® solution, so this whole scenario and our combined expertise presented an excellent opportunity. From UPM’s side, we provided the technical support and knowhow that was required from the material perspective for incorporating GrowDex into the transportation system. We were confident that it would be straightforward to encapsulate the organoids in GrowDex but had to consider the effect of plate movement during transit. GrowDex is inherently very stable so we didn’t expect any problems, even under disruptive transport conditions. However, this needed to be confirmed.”
Dr Corné Swart, Head of Business Development at Cellbox Solutions, explained the underlying principles behind the transportation system chosen for the project: “The Cellbox was developed in response to the tendency of many fragile cells, organoids and complex 3D cell cultures to fall apart and lose their structure and activity when transported. Standard cryopreservation approaches are not ideal for stem cells in particular, because cold temperatures often activate their cellular machinery and affect the reproducibility of experiments. Although adding metabolically active cryopreserving reagents can help, these additives must be removed after cells have been transported, which can cause numerous issues. The Cellbox offers a viable alternative by incubating the biological matter in CO2, mimicking natural growing conditions.”
“This pilot project presented a perfect opportunity to demonstrate the synergy of the Cellbox and GrowDex. Organoids are complex tissues that often take two or more months to grow to a stage where they can be used. Researchers cannot simply order them from a cell bank; they are very specialized systems perfected by certain groups, like the MPI, that use automated processes to reproducibly manufacture them, but they are not easily shared. Anybody currently wanting to work on them faces a real challenge to obtain them. They are very active and quite large, and consequently very sensitive to the movement and shaking they would endure during transport. And this is where the combination of GrowDex and the Cellbox came in; GrowDex provides an excellent ‘sandwich’ that keeps the organoids stabilized in an environment very similar to that in which they would normally grow. On top of this, the Cellbox systems are effectively portable battery-operated incubators with their own CO2 supplies. Of course, it’s not as simple as that, and innovative battery technology means that the Cellboxes have been developed to comply with flight legislation and logistics requirements and can be transported anywhere. We have also established good relationships with courier companies to ensure that they are handled sensitively to allow minimum disruption. We knew that these factors, together with the use of GrowDex, would protect the integrity of the cells.”
Jan described the extensive range of experiments that the project eventually included: “We started by generating two identical sets of organoids, one to be kept in the lab, and one to be shipped, and also took the opportunity to look at the effect of GrowDase™ on our tissues. GrowDase is a plant-based enzyme used to extract cells from GrowDex by digesting the matrix fibers, turning gel to liquid. Although it appears to simply dissolve the matrix away, we hadn’t investigated in detail whether the enzyme had any noticeable effect on our cells. Several conditions were set up for every eventuality: with absolutely no GrowDex, no GrowDase, some GrowDex and no GrowDase, GrowDex in different concentrations, and also GrowDase by itself. The plates were then transferred to the Cellbox, and transported by courier for eight hours before returning the cultures to the lab for testing with a couple of simple assays to measure survival.”
The project was a resounding success, with a few unexpected surprises. Jan elaborated: “The results showed that using GrowDex and the Cellbox, transport is possible without affecting cell survival, and that means we no longer need to worry about providing organoids to potential collaborators and project partners. I am confident that we can just take our cell cultures, package them into whatever format is required, and ship them to the customer. A really surprising finding was that the transported cells looked even better than the ones that were kept static in the lab. We have put this down to the fact that organoids generally like some agitation, and we predominantly perform static cell culture in the lab. Overall, these are very good results; the project itself was simple to conduct and the GrowDex and Cellbox products were very easy to use. Transporting cells, and specifically midbrain organoids, is no longer a technical challenge, and this opens up tremendous opportunities that can greatly benefit this important line of work.”