Chennakesava Cuddapah (Ph.D.), CELLnTEC Advanced Cell Systems AG, Switzerland
Introduction
Bone marrow mesenchymal stem cells (BM-MSCs), also referred to as BM mesenchymal stromal cells or marrow stromal cells, are self-renewing and expandable stem cells. The BM-MSCs are present as a rare cell population in the bone marrow, representing about 0.001% to 0.01% of the nucleated cells present. They are approximately 10-fold less abundant than hematopoietic stem cells (HSCs), are expandable in culture, multipotent and capable of differentiating into several cell types. Numerous animal studies suggest that BM-MSCs contribute to the repair/regeneration of a variety of injured tissues including the myocardium, cardiac valves, bone, tendon, cartilage, and meniscus making them a suitable candidate to use for orthopedic tissue engineering and regenerative medicine.
In this study, we investigated the suitability of GrowDex hydrogel as a matrix for 3D culture of human bone marrow-derived mesenchymal stem cells (hBM-MSC). The potential for cells to form spheroids was observed over a 7 day period and cell viability determined using AlamarBlue.
Materials
- HMSC-BM, Human Mesenchymal Stem Cells, Bone Marrow (Lifeline Cell Technology Cat # FC-0057)
- StemLife™ MSC-BM Medium Complete Kit (Lifeline Cell Technology Cat # LL-0062)
- GA Antimicrobial Supplement, 0.5 mL (Lifeline Cell Technology Cat # LS-1104)
- GrowDex 1.5 % (UPM Cat # 100 100 105)
- Low-attachment 96-well plate (Corning)
Method
- BM-MSCs were maintained according to Lifeline Cell Technology's protocol which can be found at https://www.lifelinecelltech.com.
- Cells were amplified, collected, then mixed with GrowDex and cell culture media to provide a final GrowDex concentration of 0.5 % and cell density of 1 million cells/ml.
- 65 µl of GrowDex/cell mix was dispensed into each well of a 96-well microplate and then 65 µl of culture medium carefully dispensed on top.
- The microplate was incubated at 37 °C and culture medium replaced daily.
- Cell viability was determined by adding 1/10 of the total volume (medium + hydrogel) of AlamarBlue to all wells and the plate shaken for 10 min at room temperature.
- The plate was then incubated for 3 hours at 37 °C in 5 % CO2 after which it was shaken for a further 10 min at room temperature.
- 50 μl of medium from each culture well was transferred to another 96-well plate.
- The fluorescent signal measured using a plate reader with an excitation wavelength of 480 nm and emission wavelength of 520 nm.
Results
BM-MSCs were able to quickly form spheres and establish cell function. Already after 1h from seeding it was observed that the cells started to migrate in GrowDex to form spheroids (Fig. 1A). By day 1, several spheroids were evident, having an average diameter of 140 µm (Fig 1B). Images from day 7 showed that the spheroids were starting to disaggregate (Fig. 1C). Cell viability was evaluated with AlamarBlue assay over the 7 days culture. The cells in 2D demonstrated an increased mitochondrial activity through the 7 days culture, whereas the cells in GrowDex showed an increased activity till day 1 and declined thereafter (Fig. 2).
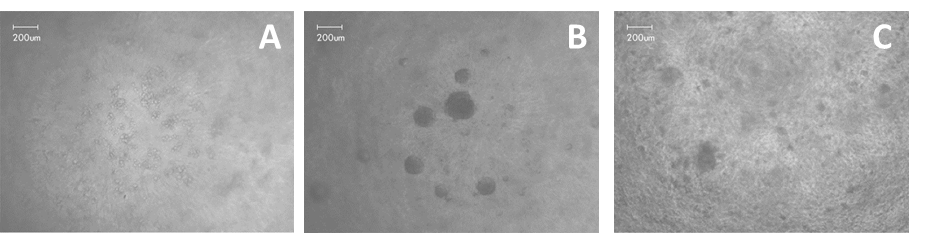
Figure 1. Human bone marrow –derived mesenchymal stem cells in GrowDex A) after 1h from seeding, B) after 24h, and c) after 7 days culture.
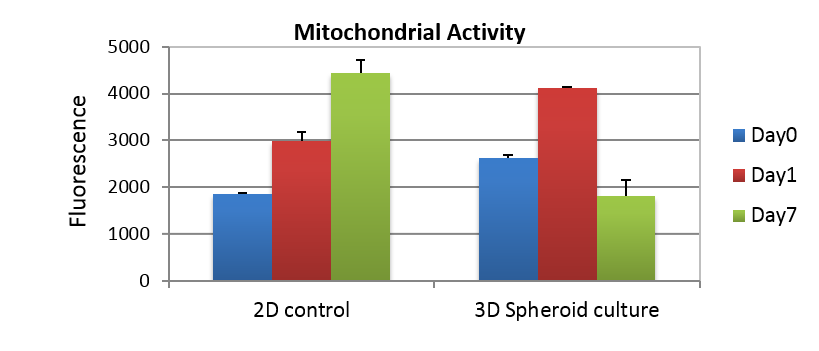
Figure 2. Cell viability (metabolic activity) determined using AlamarBlue assay studied over 7 days in 2D culture and GrowDex 3D culture.
Conclusions
Mesenchymal stem cells (MSCs) hold great promise for regenerative therapies and tissue-engineering applications. Here we have demonstrated that GrowDex is a suitable matrix for 3D culture of BM-MSCs with spheroids forming quickly after 24h in culture. The results suggest that the optimal period to use BM-MSC spheroids in GrowDex is 3-4 days after seeding.
References
Stoltz et al. 2015. Stem Cells and Regenerative Medicine: Myth or Reality of the 21th Century. Stem Cells Int. 2015;2015:734731
Rampersad 2012. Multiple Applications of Alamar Blue as an Indicator of Metabolic Function and Cellular Health in Cell Viability Bioassays. Sensors (Basel) 12(9): 12347–12360