Patrick Laurén, University of Helsinki, Finland
Introduction
Non-animal originated hydrogel nanomaterials have great potential in biomedical applications due to their versatility and soft-tissue like properties. With the ability to simulate native tissue function, hydrogels are potentially well suited for e.g. cellular therapy applications. Earlier studies with GrowDex have shown it to be an optimal culture matrix for several different cell types. In this study we investigated the rheological properties of the GrowDex-alginate hydrogels and performed cell cultures with HepG2 and SK-HEP-1 cell lines as a co-culture model within GrowDex-alginate hydrogel and on the surface, respectively. In addition, suture coating with GrowDex-alginate with HepG2 cells and performance of the coated sutures on pig liver tissue was demonstrated.
Materials
- GrowDex® (Cat No. 100 100 005, UPM)
- Sodium alginate (Cat no. W201502, Sigma), calcium chloride anhydrous (Cat No. C4901, Riedel-de-Haen), barium chloride * 2 H20 (Cat No. 529591, Sigma)
- HepG2 (HB-8065™, ATCC) and SK-HEP-1 cells (HTB-52™, ATCC)
- DMEM culture medium (Cat No. 11965092, Gibco)
- Rat collagen I (Cat No. 3443-003-01, Trevigen)
- CellTracker™ Green CMFDA (Cat No. C2925, Molecular Probes) and Red CMPTX (Cat No. C34552, Molecular Probes)
- Surgical sutures Velosorb™ Fast 3-0 (Cat No. CV916, Covidien)
Method
1. Preparation of GrowDex-alginate and co-culturing of HepG2 and SK-HEP-1 cells
a. GrowDex-alginate hydrogel was prepared by adding and mixing sodium alginate to GrowDex stock hydrogel. The mixture was left to stabilize for 24 hours. The final GrowDex-alginate mixture contained 8% (w/v) sodium alginate and 1.35% (w/v) GrowDex.
b. HepG2 cells were encapsulated in GrowDex-alginate threads by suspending the cells in GrowDex-alginate (cell density 1043cells/µl) and dispensing the mixture with a syringe and a 22G needle into 68mM calcium chloride crosslinking solution for 3 min and 20mM barium chloride solution for additional 5 min.
c. For SK-HEP-1 cell seeding, the GrowDex-alginate-HepG2 threads were treated with 1mg/ml type I collagen by pipetting collagen on the threads to cover them fully and incubating at 37°C for 30 min. The threads were transferred into low attachment culture plates, SK-HEP-1 cell suspension (1.2 million cells/ml) was introduced on top of GrowDex-alginate-HepG2 threads, and incubated at 37°C for 2h with subsequently shaking the plate every 30mins.d.The well plates were incubated at 37°C from 48h up to 2 weeks and the culture medium was replaced every 48h.
2. Coating of surgical sutures with GrowDex-alginate-HepG2 hydrogel
a. Surgical sutures (Velosorb) were coated with GrowDex-alginate-HepG2 by inserting the suture through the syringe barrel and needle orifice, filling the syringe with GrowDex-alginate-HepG2 hydrogel and feeding the suture through the needle at slow rate ensuring the formation of an even hydrogel layer with 15000 cells per cm/suture.
b. Coated sutures were treated with 68mM calcium chloride crosslinking solution for 3 min and 20mM barium chloride solution for additional 5 min.
3. Imaging of the cells
a. Cells within and on top of the threads were imaged with confocal microscopy (Leica TCS SP511 HCS A, HC PL APO10x/0.4 objective, HeNe 633nm abd DPSS 561nm lasers) with cellular dyes CellTracker™ Green CMFDA and Red CMPTX
Results
The addition of sodium alginate 8% (w/v) increased the GrowDex storage- and loss modulus contributing significantly to coating strength which was clearly seen in frequency sweeps of the hydrogels (Fig. 1). Confocal microscopy showed nearly 100% cell viability throughout the 2-week incubation period within and on the surface of the coating. Typical morphologies in the cell co-culture of spheroid forming HepG2 (inside coating) and monolayer type SK-HEP-1 (on top of coating) were observed (Fig. 2). Surgical sutures were successfully coated with GrowDex-alginate-HepG2 hydrogel, the coating remained intact during suturing operation with pig liver, and the cells remained viable (Fig. 3).
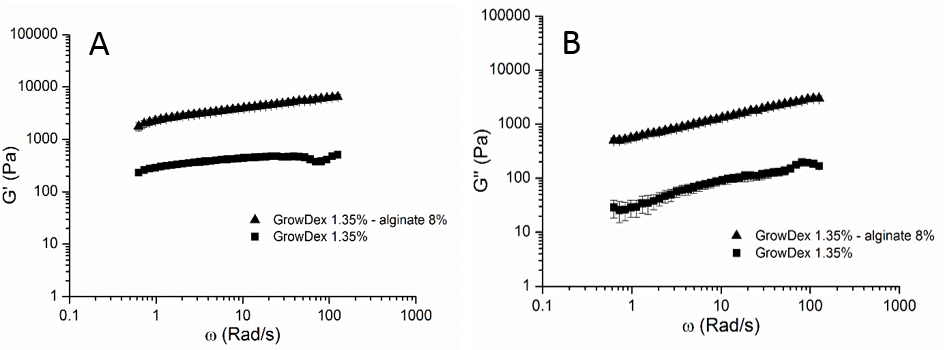
Figure 1. A) Storage modulus (G') and B) loss modulus (G’’) values showed increased strength in the GrowDex-alginate network when compared to native GrowDex.
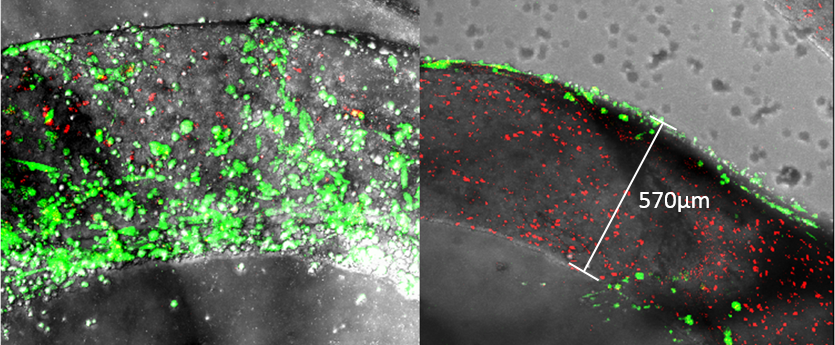
Figure 2. SK-HEP-1 cells seeded on the surface of the GrowDex-alginate threads after 48h incubation (green). HepG2 cells (red) within the threads grew individually or in very small clusters; however, surface growth showed typical cluster and epithelial morphology of both cell lines.
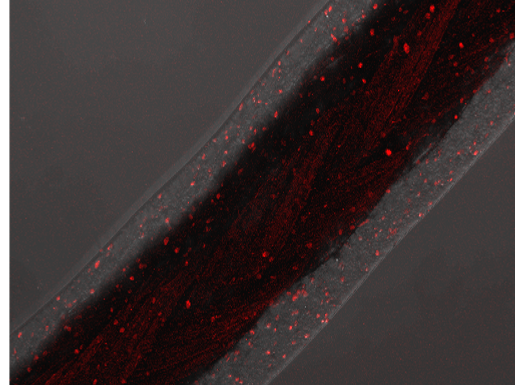
Figure 3. Surgical sutures coated with GrowDex-alginate-HepG2 and sewn three times through a pig liver segment indicating intact suture coating and live HepG2 cells within the coating matrix.
Conclusions
GrowDex-alginate hydrogel is suitable matrix for 3D co-culture of HepG2 and SK-HEP-1 cells. Cells can be cultured within and on top of GrowDex-alginate threads. In addition, surgical suture coatings from GrowDex-alginate were fabricated as biomedical devices to be used as cell-carrier systems in cellular therapy. GrowDex-alginate suture coatings could help to overcome the limitations related to cellular therapy, such as low cell survivability and cell distribution out of target tissue. Cell-containing hydrogel coated sutures can be used as cell-carrier systems for cellular based therapy and post-surgical treatment, including oral mucosa repair and ulcer treatment, and e.g. Crohn’s disease.
References
Laurén P et al. 2017 Nanofibrillar cellulose-alginate hydrogel coated surgical sutures as cell-carrier systems. PLoS ONE 12(8)