Andrew Schwab, PhD
ORISE Contractor, Research Triangle Park, NC, USA
INTRODUCTION
Three-dimensional (3D) in vitro culture of neuronal cells is necessary to recapitulate the complex cytoarchitecture of the central nervous system. As complex, multicellular models develop, there is a need for defined substrates that can allow for the differentiation of iPSC-derived neurons and astrocytes. Here we establish methodologies for 3D cell culture of human iPSC-derived neural progenitors within nanofibrillar cellulose (NFC) hydrogel, GrowDex®-T.
MATERIALS
- DMEM/F12 media (Cat No. 11330032, ThermoFisher)
- B27 Supplement (Cat No. 17504044 , ThermoFisher)
- GrowDex-T, 1.0% (Cat No. 200 103 005, UPM)
- 96-well plate (Cat No. 89626, Ibidi)
- Anti βIII Tubulin mAB (Cat No. G7121, Promega)
- Anti GFAP mAB (Cat No. 12389, Cell Signalling Technology)
- Matrigel (Cat No. 354277, Corning)
METHODS
- Human induced pluripotent stem cell (iPSC) derived neural progenitors (1) were cultured for one week in GrowDex-T and on Matrigel.
- Studied GrowDex-T concentrations were 1, 0.8, 0.6, 0.4 and 0.2 wt%.
- GrowDex-T hydrogel volume was 100 µL in a Ibidi 96-well plate.
- Matrigel volume was 100 µL and used at a final concentration of 0.08 mg/mL. The media was aspirated and the cells were plated on top of the Matrigel layer.
- Cell concentration for all conditions was 50,000 cells per well.
- Cells were fed every other day with DMEM/F12 containing 2% B27.
- Half of the media was replaced as to not disturb the hydrogel layer.
- The formation of neural cells inside the hydrogels was evaluated by immunocytochemical staining against βIII Tubulin and GFAP.
RESULTS
After one week of differentiation in GrowDex-T and Matrigel, neural cells were observed by immunotcytochemistry. Representative 10x images of neurons and astrocytes are shown in Fig. 1. Qualitatively, neural growth increased as GrowDex-T concentration increased from 0.2% to 1% (Fig. 1). GrowDex-T appeared to support increased neural growth and neurite elongation as compared to previous results with native GrowDex (2). Although, aspiration induced cell loss in GrowDex conditions may have attributed to this difference, as neural cells grown in GrowDex were more susceptible to detachment from the plate.
Cultures in GrowDex-T resulted in 3D neural structures and outgrowth, whereas cells on Matrigel resulted in neuronal monolayer.
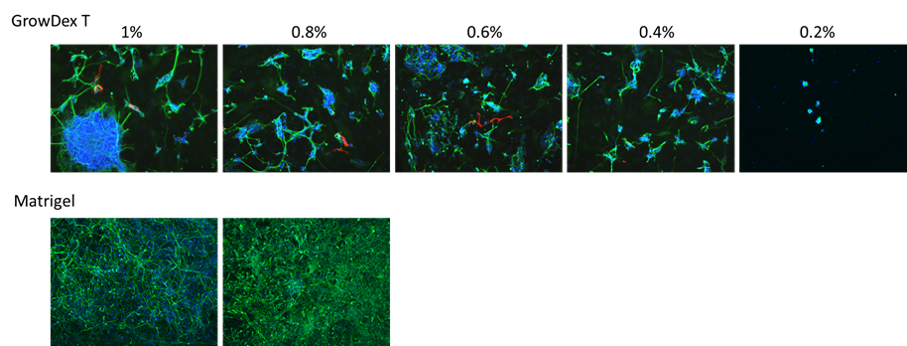
Figure 1. EZ spheres differentiate into neurons and astrocytes within GrowDex-T. Representative images showing cell nucleai (blue), differentiation of neural progenitors into βIII Tubulin positive neurons (green), GFAP positive astrocytes (red) in GrowDex-T and Matrigel (2 representative images).
CONCLUSIONS
Compared to 2D, differentiation of neural progenitor cells in a 3D environment represents a more physiological approach to model organ-level responses. Neural progenitors grown on Matrigel differentiated into a monolayer of neurons whereas neural progenitor cells grown in GrowDex-T resulted in neuronal 3D structures of neurons and astrocytes. Based on the results, it can be concluded that GrowDex-T is a good substrate for 3D culture of human iPSC-derived neural progenitor cells. The biocompatible nature of GrowDex-T and ease of use in a 96-well plate format provides a promising alternative scaffold for 3D cell culture. Transparency of GrowDex-T hydrogel enables excellent imaging quality.
Notably, these results demonstrate that GrowDex-T could easily be adapted for use in 3D microfluidic neural cell culture models where a defined, animal-free, and tunable matrix may be required. As 3D cultures continue to grow, GrowDex-T will be an important tool that may improve the success of new alternative models.
REFERENCES
1.Ebert, A.D., et al., EZ spheres: a stable and expandable culture system for the generation of pre-rosette multipotent stem cells from human ESCs and iPSCs. Stem Cell Res, 2013. 10(3): p. 417-427.
UPM Biomedicals Application Note AN027 ” GrowDex® Supports Human iPSC-derived Neural Progenitor Differentiation in vitro”