Dr. Janina Noy and Dr. Robert Utama (Inventia Life Science, Australia) and
Dr. Markus Nuopponen (UPM, Finland)
INTRODUCTION
Three-dimensional (3D) cell culture has been established to be more accurately representing in vivo physiological conditions than 2D cell cultures [1]. RASTRUM 3D bioprinting platform (Fig. 1) by Inventia Life Science enables high throughput creation of 3D cell models, in an efficient and reproducible way. RASTRUM utilizes digital bioprinting technology that prints tiny droplets of cells and matrix components in a way that is very fast, very precise and gentle on cells. RASTRUM utilizes advance bioink technologies, such as nanofibrillar cellulose based bioink GrowInk™-T (UPM, Finland), to print 3D cell models which closely resemble the extracellular matrix and supports cell growth in 3D.
GrowInk-T is an animal free, biocompatible, ready to use hydrogel bioink that can be mixed directly with cells for bioprinting applications. It is shear-thinning material that allows easy printing. GrowInk-T mimics the in vivo environment supporting cell growth and differentiation and can be diluted to provide a wide range of viscosities that can be matched to the requirements of the cells. Equipped with an inbuilt laminar flow hood, RASTRUM 3D bioprinting platform is a complete solution to create biologically relevant 3D cell models with unprecedented speed and reproducibility.

MATERIALS
- Human breast (adenocarcinoma) cell line (MCF-7, ATCC HTB-22)
- GrowInk-T (UPM, Finland) diluted 1:1 with ultra pure water, and further diluted 3:2 in PBS
- 96-well plate (Thermo Fisher Scientific)
- DMEM culture media (Thermo Fisher Scientific)
- Foetal Bovine Serum (FCS) (Bovogen)
- PrestoBlue Cell Viability Reagent (Thermo Fisher Scientific)
- RASTRUM print cartridge (Inventia Life Science)
METHODS
1.RASTRUM platform preparation
- RASTRUM bioprinter was initialized to start the laminar flow hood. Bioprinter surfaces were sterile wiped with 70 v/v% ethanol.
- To prepare the printing cartridge, 200 μl of diluted GrowInk-T was pipetted into a sterile RASTRUM print cartridge, pre-filled with the sterilization fluids. The cartridge and a sterile well-plate were placed into their respective designated slots in the printer.
- RASTRUM bioprinting
- The bioprinting process was started with the automated fluidics sterilization process. Briefly, the fluidics were flushed with 70 v/v% ethanol, followed by sterile DI water.
- After the sterilization phase, the printer automatically primed 200 µl of GrowInk-T
- To assess the reproducibility of the bioprinting process, multiple 3D structures as shown in Fig. 2 were bioprinted on a 96-well plate.
- At the completion of the bioprinting process, an automated shutdown cleaning process was conducted with 70 v/v% ethanol and sterile DI water, to maintain the sterility and reliability of the bioprinter.
-
-
3.Cell viability investigation in GrowInk-T
- One million MCF-7 cells were harvested and resuspended homogeneously in 100 μl GrowInk-T, to yield a cell suspension at 10 million cells/ml.
- 15 μl of the cell-GrowInk-T suspension was transferred into 5 centrifuge tubes.
- Every 15 min, 1 ml DMEM was added to one of the tubes. 20 μl of the DMEM cell-GrowInk-T suspension was then plated into a 96-well plate and topped up with 80 μl DMEM/10% FCS.
- This process was repeated 4 times after 15, 30, 45 and 60 min of incubating the MCF-7 suspension in GrowInk-T at room temperature.
- 100 μl of DMEM/10% FCS was used as blank control.
- The plate was incubated for 24 h and a PrestoBlue viability assay was performed according to the manufacturer’s protocol.
RESULTS
RASTRUM 3D bioprinting of GrowInk-T
We combined RASTRUM 3D bioprinter with Growink-T biomaterial to validate a new capability in generating biologically relevant 3D cell models reproducibly and in high-throughput. 3D cuboid structure was selected as the model structure as it is currently used to form embedded 3D spheroids and organoids for drug screening application. The printed cuboid measured 1.5 mm in length and 300 µm in height, with a volume of ~500 nl (Fig. 2). The resulting hydrogel fits in the 10x objective field of view, clear in appearance and has a compatible thickness for fluorescence-based downstream analysis.
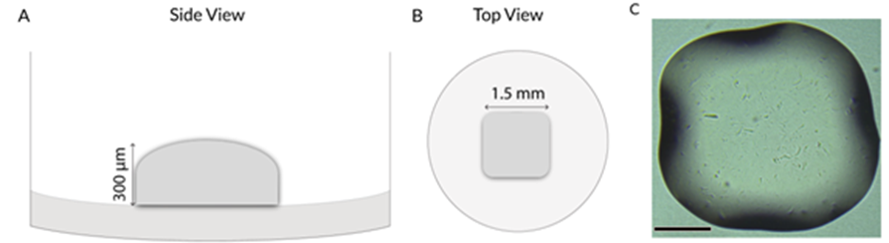
Figure 2. Representative 3D bioprinted GrowInk-T plug in a 96-well plate. A) Schematic side view of the cuboid structure, 300 μm in height, B) Schematic top view of plug, 1.5 mm in diameter, C) Bright-field image of the cuboid structure, Scale bar: 500 μm.
To demonstrate the reproducibility of the bioprinting process, we ran an automated printing process of a full 96-well plate containing the cuboid structure. A full well plate was printed in less than 10 min. The well plate was subsequently imaged and analyzed for variability (Fig. 3). Each well was confirmed to have a structure with the same size and shape definition.
Moreover, the cuboid structure was printed at a consistent location across all wells. Both analyses confirmed that not only the ejection of the droplets were consistent in each structure, but also across all wells. The printing process was also repeated twice, with each print utilizing a freshly prepared GrowInk-T bioink. The same outcomes were observed in those samples, indicating a strong batch-to-batch consistency of the printing and the bioink. In total 182 wells of structures were printed with no variation in the structure definition and location within a well, attributes that are beneficial for automated high-content imaging.
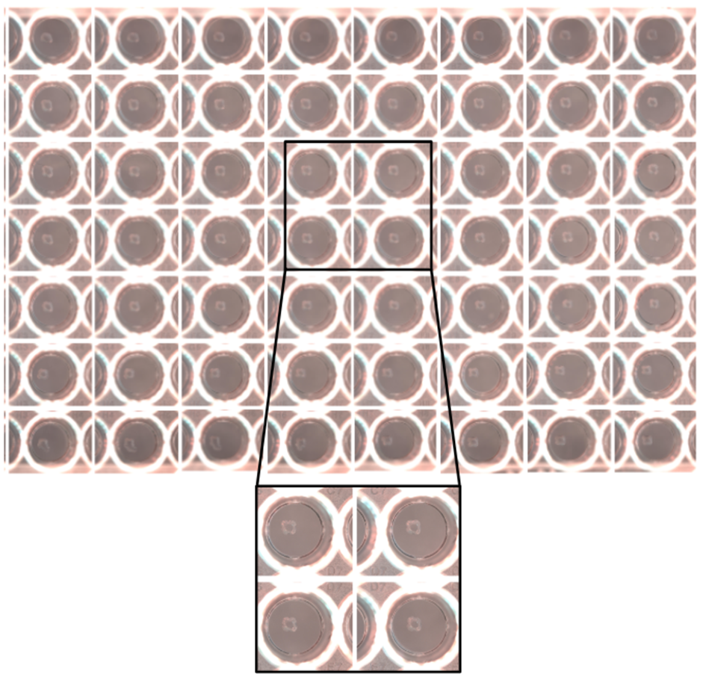
Figure 3. Representative GrowInk-T print in a 96-well plate.
It is crucial to maintain high cell viability while the cells are suspended in the bioink during the printing process. We used MCF-7 as the model cell as previous reports have shown that MCF-7 cells form well-defined 3D spheroid structures in nanofibrillar cellulose hydrogel, GrowDex®[2]. To mimic the environment that MCF-7 cells were exposed to when suspended in the bioink during bioprinting, MCF-7 cells were suspended at 10 million cells/ml in 100 µl of GrowInk-T. The cell suspension was incubated at room temperature for 60 minutes with samples collected every 15 min. Subsequent PrestoBlue analysis of the samples after 24 h incubation at 37°C showed a viability of 98% for the printing duration (< 15 min), while still maintaining a high cell viability of 88% after 60 min suspension in the GrowInk-T (Fig. 4).
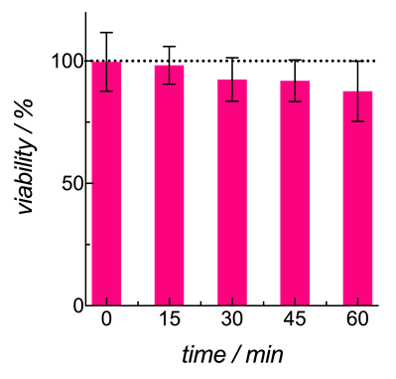
Figure 4. MCF-7 viability when suspended in GrowInk-T over a period of 60 minutes.
CONCLUSIONS
RASTRUM 3D bioprinting platform was successfully utilized to bioprint GrowInk-T hydrogel. A full 96-well plate of 3D structure was printed in 10 min. Bioprinted 3D structures were shown to be reproducible from well-to-well, plate-to-plate and batch-to-batch. MCF-7 cell viability studies over 60 min showed that the bioink is biocompatible for cell suspension. By combining RASTRUM with the validated cell applications that GrowInk-T enables, in vitro 3D cell culture models of both cancerous and primary cells can be produced fast, efficiently and reproducibly, suitable for high content screening applications.
REFERENCES
[1] Haycock J.W. (2011) 3D Cell Culture: A Review of Current Approaches and Techniques. In: Haycock J. (eds) 3D Cell Culture. Methods in Molecular Biology (Methods and Protocols), vol 695. Humana Press.
[2] Niklander, J. and Paasonen, L. “Breast Cancer Cell Line MCF7 3D Culture Demonstrated in GrowDex® and Matrigel”, UPM Biofore Application Note 29.