Piia Mikkonen 1, Aino Palva 2, Pekka Ellonen 2 and Vilja Pietiäinen 1
1 Institute for Molecular Medicine Finland-FIMM, Helsinki Institute for Life Sciences-HiLIFE,
University of Helsinki (UH), Finland
2 FIMM Sequencing Unit, FIMM Technology Center, Institute for Molecular Medicine Finland,
Helsinki Institute for Life Sciences-HiLIFE, University of Helsinki, Finland
Introduction
In the individualized systems medicine, patient-derived cancer cells (PDCs) are utilized ex vivo to study the drug responses at the individual patient level (Pemovska et al., 2013; 2015; Pietiäinen et al., 2014). When studying solid tumors, 3D cell culture conditions are considered to be physiologically relevant as they may resemble the tissue surrounding the tumor cells (Horvath et al., 2017). Importantly, the PDCs grown in 2D or 3D cultures should be genetically characterized to confirm that they carry the same cancer driver mutations as the original tumor they were derived from (Saeed et al. 2017; 2018). To this aim, for example exome or panel sequencing is performed to germline sample (blood), tissues and PDCs. Typically, the DNA isolation is performed from PDCs using commercial column extraction methods. However, the properties of many 3D matrices may not allow the proper filtration of samples through the DNA isolation column, resulting in an insufficient coverage of the DNA. We tested in this proof-of-concept study a direct recovery of DNA from cells grown in GrowDex® after degradation of the matrix with GrowDaseTM cellulase enzyme.
Here, we describe a streamlined workflow for constructing an NGS whole exome sequencing (WES) from a low amount of PDCs grown as spheroids in GrowDex.
Materials
- GrowDex®, 1.5% (Cat No. 100 103 005, UPM)
- GrowDaseTM, 10 mg/ml (Cat No. 900 102 002, UPM)
- Ultra Low Attachment (ULA) 24-well plates (Cat No. 3473, Corning)
- Dynabeads® DNA DIRECT Universal kit (Cat No. 63006, Thermo Fisher Scientific)
- KAPA Hyper Plus kit (Cat No. KK8514, Roche Molecular Systems Inc.)
- Roche single index adapters, Medexome probes and HyperCap 2.0 protocol (Roche Molecular Systems Inc.)
- HiSeq2500 system (Illumina)
- 2100 Bioanalyzer High Sensitivity assay (Agilent)
- Qubit BR System (Thermo Fisher Scientific)
Methods
- PDCs were dissociated from renal cancer/benign tissue samples (Saeed et al., 2017; 2018). The dissociated cells were resuspended to 600 µl of the complete growth medium including 0.2% and 0.4% GrowDex, and seeded to 24-well plates.
- GrowDase was diluted to working concentration of 600 µg/mg (1.2 mg/ml for GrowDex 0.2% and 2.4 mg/ml for GrowDex 0.4%) with culture medium and 600 µl was added to GrowDex culture, mixed 5 times with a pipette and the plate was incubated overnight at 37°C. The content of wells was collected by centrifugation (1000 x g, 5 minutes in RT) and processed for the DNA recovery and NGS library construction. The cell counts for the DNA isolations varied from 20 000 to 100 000 cells.
-
The Dynabead DNA Direct reagent was mixed to 1:1 with cell suspension and DNA concentration was measured from the bead-bound samples using Qubit. DNA library construction was performed without eluting the DNA off the beads.
-
Enzymatic DNA fragmentation approach was chosen for the NGS library construction. NGS libraries were constructed using the KAPA Hyper Plus kit and exome capture was performed with Roche MedExome probes following the Roche HyperCap 2.0 protocol.
-
150 ng of DNA was used for the library construction. Roche single index adapters were used for barcoding the libraries. Ligation time was 1 hour at 20 °C. Seven cycles of PCR were performed in the library construction and 12 cycles in the post capture PCR. A total of 1000 ng consisting of 3-4 barcoded NGS libraries were pooled together for each capture. Library size distribution and molarity were determined by Agilent 2100 Bioanalyzer High Sensitivity assay.
-
Sequencing was performed with Illumina HiSeq2500 system in Rapid mode using HiSeq Rapid v2 kits. Read length for the paired-end run was 2x101 bp. Sequencing results were analyzed using a variant calling pipeline (VCP) developed in FIMM, UH.
Results
The PDCs formed spheroids of 60-120 microns in GrowDex (Fig. 1A) in 24-well plates. After GrowDase treatment, the hydrogel was degraded but the spheroids stayed intact (Fig. 1B).
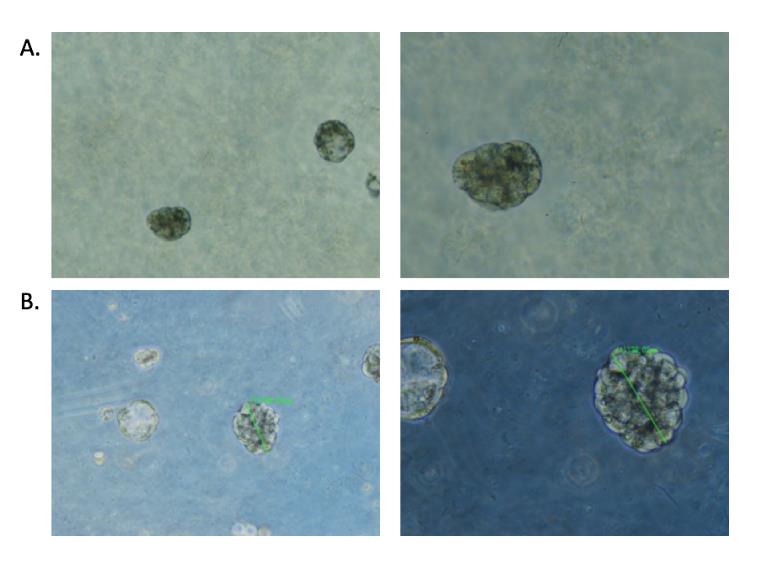
Figure 1. PDC spheroids in GrowDex (A) and after overnight degradation of GrowDex with GrowDase enzyme (B). Left panel 5X objective; Right panel 10X objective.
GrowDase treatment was crucial for the successful bead-based isolation as the cell-bead-complex was difficult to handle in the absence of the treatment. GrowDase treated spheroids combined with beads were robustly processed on the magnetic rack. The yields of the bead-based direct isolations varied from 200 to 600 ng of DNA.
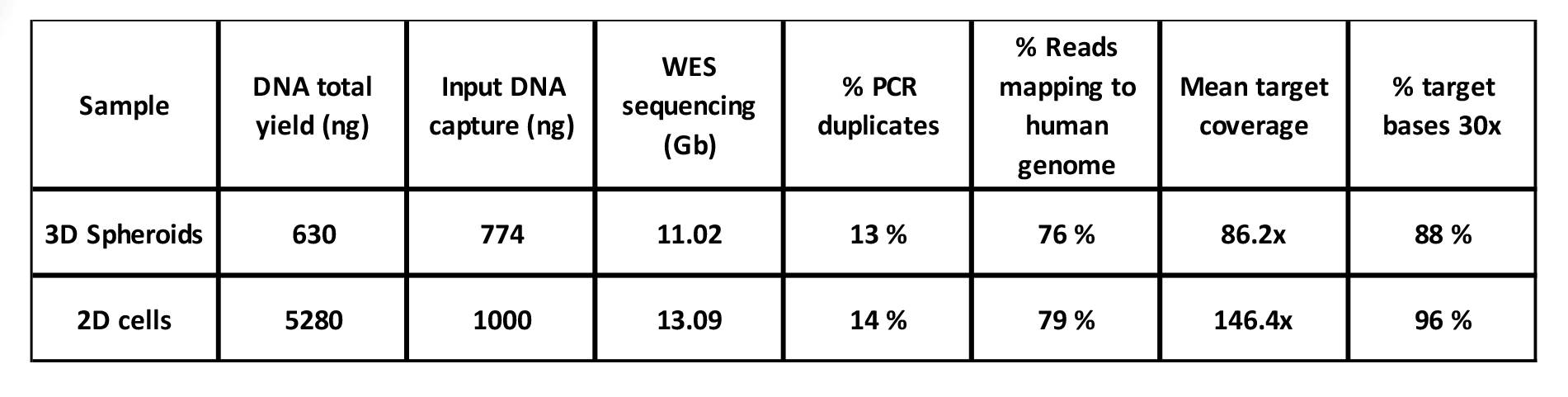
The success of exome capture is critically dependent on the amount of input DNA. This may have caused variation in the results. While in general the good yield of DNA was obtained from samples (Table 1), the yield of the library before exome capture was not equally high in all of the libraries, varying from 200 to 774 ng of DNA (data not shown). The results were comparable with 2D culture of the same sample. The applying of the sample immediately after GrowDase treatment to Dynabead reaction may improve the results, and the effect of longer storage of sample at 4°C prior to DNA isolation should be systematically tested.
Table 1. NGS analysis statistics of libraries from PDCs grown either in 2D or in 3D in 0.4% GrowDex.
To gain more molecules suitable for amplification, the ligation time was prolonged from 15 minutes to an hour. PCR cycles remained the same throughout the optimizing of reactions. Here, overamplification was observed with some samples with PCR duplication rates as high as 66% of the library, suggesting that the ligation conditions and PCR cycles may need further optimization when a low amount of input DNA is available. This could help to gain enough diversity for the sample library.
The comparison of sequencing results revealed the similar coverage in the sequencing of DNA isolated from PDCs cultured either in 2D on plastic or 3D in GrowDex as exemplified in Fig 2.
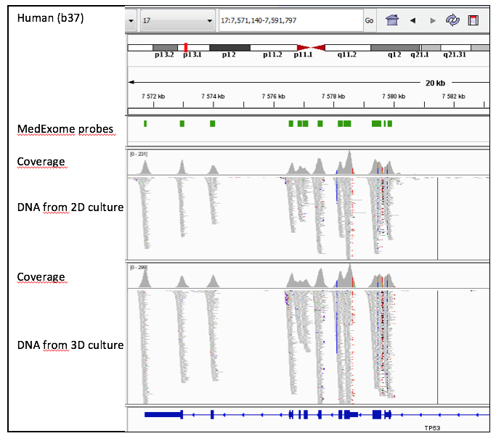
Figure 2. A snapshot image from the IGV (the Integrative Genomics Viewer (IGV; Robinson et al., 2011; Thorvaldsdóttir et al., 2013) of analysed data shows the similar coverage of TP53 sequencing (chr 17) in DNA from 2D and 3D cultured PDCs.
Conclusions
Our proof-of-concept study introduces a method to efficiently isolate DNA from PDCs grown as spheroids in GrowDex. The enzymatic degradation of GrowDex with GrowDase makes the cells accessible for lysis and bead-based DNA recovery. Recovered amount and quality of DNA is sufficient for NGS library construction, when enzymatic DNA fragmentation is used in combination with extended ligation times. Protocol may require further optimization depending on the amount of cells used and GrowDex concentration.
This straightforward protocol omits additional purification steps typically present in NGS library construction, which is important for characterization of PDCs grown in 3D conditions in GrowDex.
References
Horvath P, Aulner N, Bickle M, Davies A, Del Nery E, Ebner D, Montoya M.C, Östling P, Pietiäinen V, Price L, Shorte SL, Turcatti G. von Schantz-Fant C, and Carragher NO, the EuCAI (European Cell Based Assay Interest Group). Screening out irrelevant cell-based models of disease, Nat Rev Drug Discov. 2016 Nov;15(11):751-769.
Pemovska T, Kontro M, Yadav B, Edgren H, Eldfors S, Szwajda A, Almusa H, Bespalov MM, Ellonen P, Elonen E, Gjertsen BT, Karjalainen R, Kulesskiy E, Lagström S, Lehto A, Lepistö M, Lundán T, Majumder MM, Lopez Marti JM, Mattila P, Murumägi A, Mustjoki S, Palva A, Parsons A, Pirttinen T, Rämet ME, Suvela M, Turunen L, Västrik I, Wolf M, Knowles J, Aittokallio T, Heckman CA, Porkka K, Kallioniemi O, Wennerberg K. Individualized Systems Medicine (ISM) strategy to tailor treatments for patients with chemorefractory acute myeloid leukemia. Cancer Discov. 2013 Dec;3(12):1416-29.
Pemovska T, Johnson E, Kontro M, Repasky GA, Chen J, Wells P, Cronin CN, McTigue M, Kallioniemi O, Porkka K, Murray BW, Wennerberg K. Axitinib effectively inhibits BCR-ABL1 (T315I) with a unique binding conformation. Nature 2015, 519(7541):102-5, 2015.
Pietiäinen V, Saarela J, von Schantz-Fant C, Turunen L, Östling P, and Wennerberg K. The High Throughput Biomedicine unit at the Institute for Molecular Medicine Finland: High throughput screening meets precision medicine. Combinatorial Chemistry and High Throughput Screening 2014 17(4):377-86.
Robinson JT, Thorvaldsdóttir H, Winckler, W, Guttman M, Lander ES, Getz G, and Mesirov JP. Integrative Genomics Viewer. Nature Biotechnology 2011 29, 24–26.
Saeed K, Rahkama V, Eldfors S, Bychkov D, Mpindi JP, Yadav B, Paavolainen L, Aittokallio T, Heckman C, Wennerberg K, Peehl DM, Horvath P, Mirtti T, Rannikko A, Kallioniemi O, Östling P, Af Hällström TM. Comprehensive drug testing of patient-derived conditionally reprogrammed cells from castration-resistant prostate cancer. Eur Urol. 2017 Mar;71(3):319-327.
Saeed K, Ojamies P, Pellinen T, Eldfors S, Turkki R, Lundin J, Järvinen P, Nisen H, Taari K, Af Hällström TM, Rannikko A, Mirtti T, Kallioniemi O, Östling P. Clonal heterogeneity influences drug responsiveness in renal cancer assessed by ex vivo drug testing of multiple patient-derived cancer cells. Int J Cancer. 2018 Aug 19. (epub ahead of print).
Thorvaldsdóttir H, Robinson JT, and Mesirov JP. Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Briefings in Bioinformatics 2013 14, 178-192.