Beate Rinner, Medical University of Graz, Austria, Johanna Niklander, Raili Koivuniemi and Marjo Yliperttula, University of Helsinki, Finland
Introduction
NRAS mutation in melanoma is associated with aggressive tumor biology and poor prognosis. Therapy response rates for NRAS mutated melanoma are significantly weaker than for BRAF mutated melanoma. Well-characterized NRAS mutated cell lines with correct phenotype are needed for research and drug development. While plenty of cell lines exist, only few melanogenic cell lines retain their in vivo characteristics.
In this application note, 3D culture of novel NRAS mutated human melanoma cell line MUG-Mel2 in GrowDex® is presented. MUG-Mel2 was derived from a patient with aggressive cutaneous ulcerated melanoma. The 3D culture matrix was required to be tuned for MUG-Mel2 due its novel status, and the scaffold needed also to be removable for further downstream analysis. In previous studies GrowDex has been shown to be suitable 3D culture matrix for various cell types, supporting also organotypic growth, due to the tunability of the mechanical properties of the hydrogel. GrowDex can be also removed by simple cellulase enzyme treatment.
Materials
- MUG-Mel2 cell line (Medical University of Graz)
- RPMI cell culture medium with 10% FBS, 2mM L-glutamine, 1% penicillin/ streptomycin (Cat No. 11875-093, Life Technologies)
- GrowDex® (Cat No. 100 300 105, UPM)
- GrowDase™ (Cat No. 900 100 102, UPM)
- Cellstain double staining kit (Cat No. 04511, Sigma)
- DAPI (Cat No. D1306, Invitrogen)
- Prolong Diamond Antifade Mountant (Cat No. P36965, Life Technologies)
Method
- To determine the optimal conditions for 3D cell culturing, three concentrations (0.4 %; 0.7 % and 1 % wt/v) of GrowDex, as well as two cell-seeding concentrations (50,000 and 100,000 cells per 100 µl of GrowDex/96-well) were tested.
- MUG-Mel2 spheroids grown in GrowDex were collected at day three for live calcein staining. Prior to collection, GrowDex was degraded from the cell culture by GrowDase (UPM) cellulase enzyme treatment for 24 h at 37 °C using 300 µg GrowDase per 1 mg of cellulose in GrowDex.
- Confocal imaging:
a.Spheroids were washed three times with 1xDPBS, suspended in staining solution containing Calcein-AM 1:500 (Cellstain double staining kit, Sigma) and 50 µg/ml DAPI (Invitrogen) for 15 min at 37 °C, and subsequently mounted using Prolong Diamond Antifade Mountant (Life Technologies).
b.Fluorescent images were taken with the Leica TCS SP5 microscope using HCX PL APO 20x/0.7 objective.
Results
MUG-Mel2 cells were able to form multicellular spheroid structures after one day culture in GrowDex. The optimal condition for the spheroid formation was found to be between 0.4 % and 0.7 % GrowDex concentration with cell seeding density of 50,000 cells/ml. In 1 % GrowDex MUG-Mel2 cells did not form spheroids. After three days in culture, MUG-Mel2 formed spheroids with diameter around 200 µm, showed good viability and correct morphology (Fig. 1 A-C). High pigmentation typical for melanoma cells was clear after five days culture in GrowDex (Fig. 1D).
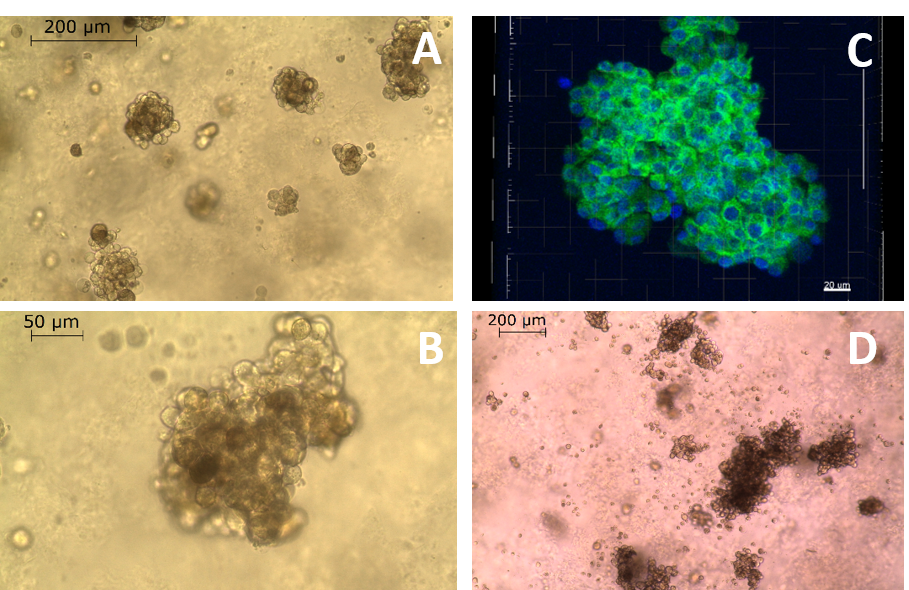
Figure 1. Morphology of MUG-Mel2 cell spheroids in GrowDex after three days culture in a concentration of 0.5 % (A,B). Calcein staining (green) revealed the viability of the spheroids, nuclei were counterstained with DAPI (blue) (C). After five days in GrowDex, MUG-Mel2 showed intensive brown staining of the cell cytoplasm typical for melanoma cells (D).
Conclusions
The newly established cell line MUG-Mel2 cultured in GrowDex offers a promising model for NRAS mutated melanoma research and therapy development. GrowDex presents an optimal matrix for the cells to maintain their natural behavior and the unique phenotype of the melanoma - the stable brown coloring. Cell culture models that maintain characteristics of the in vivo state are important research models to explore new and relevant targets for melanoma treatment.
References
Rinner B. et al. 2017 MUG-Mel2, a novel highly pigmented and well characterized NRAS mutated human melanoma cell line. Scientific Reports 2017; 7, 2098.