Hsiang-Tsun Chang1, Rachel A. Heuer1, Andrew Oleksijew1, Kyle S. Coots1, Christian B. Roque1, Shun Kobayashi1, Duncan M. Chadly1, Kevin T. Nella1, Tony Kiuru5, Lauri Paasonen5, Jonathan Sheard5 and Akihiro J. Matsuoka1,3,4,
1 Department of Otolaryngology-Head and Neck Surgery
² Department of Neurology
3 Department of Communication Sciences and Disorders
4 Knowles Hearing Center, Northwestern University, Chicago and Evanston, IL, U.S.A.
5 UPM Biomedicals, Helsinki, Finland
INTRODUCTION
In 2018, the WHO reported that sensorineural hearing loss (SNHL) affects 6.1% of the world’s population (466 million people). This is estimated to rise to 900 million people in 2050 [1]. Stem cell therapy is a promising method to alleviate SNHL, but its potential is currently hindered by low cell survival post transplantation [2-5]. The harsh, nutrient deprived environment of the cochlea is thought to be a leading factor in poor cell survival [6, 7]. Therefore, for human embryonic stem cell (hESC) derived otic neuronal progenitors (ONP) to survive in the inner ear, a suitable stem cell niche is needed. To alleviate this problem, we have evaluated the use of GrowDex®-T in a 3D cell culture setup, to establish a robust and reproducible in vitro assay whilst developing an artificial stem cell niche. GrowDex-T was used as an artificial extracellular matrix for the delivery of hESC-derived ONP spheroids along with brain derived neurotrophic factor (BDNF) PODS™-crystals into the mouse inner ear.
Keywords: GrowDex-T, SNHL, hESC, Hearing Loss, ONPs, PODS™
MATERIALS
- Human embryonic stem cells, specifically H1, H7 and H9 (WiCell Madison, WI, USA)
- Neuronal induction medium (NIM): Neurobasal medium (#21103049; Thermo Fisher Scientific, Waltham, MA, USA), 1% N2 Supplement-A, 2% B27 Supplements, and 1 mM Glutamax™ Supplement (#35050061, Thermo Fisher Scientific, Waltham, MA, USA)
- Clear Round Bottom Ultra-Low Attachment Microplate® (Cat No. 7007, Corning Life Science, USA)
- EZSPHERE™ culture plate (Cat No. 264170, Nacalai)
- Flat Bottom Clear Plate® (Greiner Bio-One, Germany)
- GrowDex-T, 1.0% (Cat No. 200 103 005, UPM Biomedicals, Finland)
- Recombinant human brain derived nerve factor (RhBDNF), 20ng/ml (R&D system, Minneapolis, MN, USA)
- PODS™ Human BDNF(PODS-hBDNF) (Cat No. PPH1-250, Cell Guidance Systems)
- BrainPhys™ Neuronal Medium (Cat No. 05790, Stem Cell Technologies) with 10 % FBS.
- Blocking solution: 5% bovine serum albumin (Sigma-Aldrich, USA) and 0.1% Triton X-100 in DPBS (Corning USA)
- Immunocytochemistry (ICC) staining reagents:
- Primary antibodies: PAX8 1:100, β-III tubulin 1:1,000 (Cat No. ab13611 and Cat No. ab18207 respectively, Abcam, USA)
- Secondary antibodies: Alexa Fluor 488 and 647 at 5 µg/ml (Cat No. 150129 and Cat No. 150075, respectively, Abcam)
- DAPI (Cat No. D1306, Thermo Fisher Scientific, USA)
- Nikon A1 confocal microscope (Nikon, Minato, Tokyo, Japan)
- ImageJ ver. 2.0.0-rc-69/1.52p. (National Institutes of Health, Bethesda, MA, USA)
METHODS
- Human embryonic stem cell (hESC) derived late-stage otic neural progenitors (ONPs), as previously described and characterized [8], were dissociated into single cells from a traditional monolayer plate and transferred for sphere formation into either:
- 96-well clear round bottom ultra-low attachment microplate® (Corning) at 5x104 cells/well
- EZSPHERE® culture plate (Nacalai) that consists of uniformly sized microwells coated with a low-binding affinity polymer (2-methacryloylxyethyl phosphorycholine) at 1x106 cells/plate
- More detailed steps can be found in supplementary data presented in Chang, H.-T. et al., (2020).Cells were cultured in an ONP maintenance media (ONPMM) (7) at 37°C, 5% CO2 for two days and allowed to form spheroids with diameters of roughly 250 μm.
- GrowDex-T stock was diluted to final concentrations of 0.375% (v/v) or 0.5% (v/v) with PBS, RhBDNF (20 ng/ml) or 8x105 PODS-hBDNF (equal to 20 ng/ml).
- Working example for preparation of 1 ml of 0.5% GrowDex-T with PODS: reconstitute PODS-hBDNF at 2x108 co-crystals/ml in water. Add 4 µl to 496 µl of PBS within a microcentrifuge tube. Add 500 µl of GrowDex-T and mix thoroughly.
- 50 μl of diluted GrowDex-T with PODS-hBDNF was plated into a well of a 96-well Flat Bottom Clear Plate.
- Spheroids were carefully transferred into the GrowDex-T by direct injection using a P200 micropipette tip, then 500 μl of BrainPhys was gently added on top of the GrowDex-T.
- Spheroids were cultured for 7 days at 37°C, 5% CO2 with no media
- Following 7 days, the media was removed by ensuring the pipette tip was above the cells and GrowDex-T layer. Then the cells and GrowDex-T were fixed with 4% PFA and fluorescently stained for Peripherin, VGLUT2, and
- ICC staining of spheroids:
- All culture media was carefully removed from the top of the well without disturbing the GrowDex-T and then the embedded spheroids were fixed with 4% (w/v) PFA for 30 mins. Spheroids in GrowDex-T were washed 2-3 time with PBS then incubated overnight in blocking solution.
- Each sample was then incubated for three days at room temperature with the primary antibodies followed by several wash steps with PBS. Samples were then incubated with the secondary antibodies in the dark for three days at room temperature.
- Nuclei were then stained by incubating samples with DAPI for 30 minutes. After incubation with DAPI, all samples were washed and maintained in PBS.
- Images if were acquired with a Nikon A1 confocal microscope (Nikon, Tokyo, Japan). ImageJ was used to quantify the number of immune-positive cells, neurite length, bearing and arborization from the images. Further details on image acquisition and quantification of are described in the Supplementary Data.
- From the acquired images, the number of PAX8 positive spheroids were quantified, along with measurements of neurite length, bearing and arborization. *Further details can be found in the Supplementary Data.
RESULTS
hESC derived ONP spheroids were successfully cultured in GrowDex-T with PODS-hBDNF (Figure 1, A) for seven days. It was noted that the spheroids, when cultured in GrowDex-T alone, maintained their shape and integrity from 24 through 72 hours (Figure 1, B a-c), whilst the addition of PODS-hBDNF acted as a chemo-attractant for the spheroids (Figure 1, B d-f) and induced them to further differentiate.
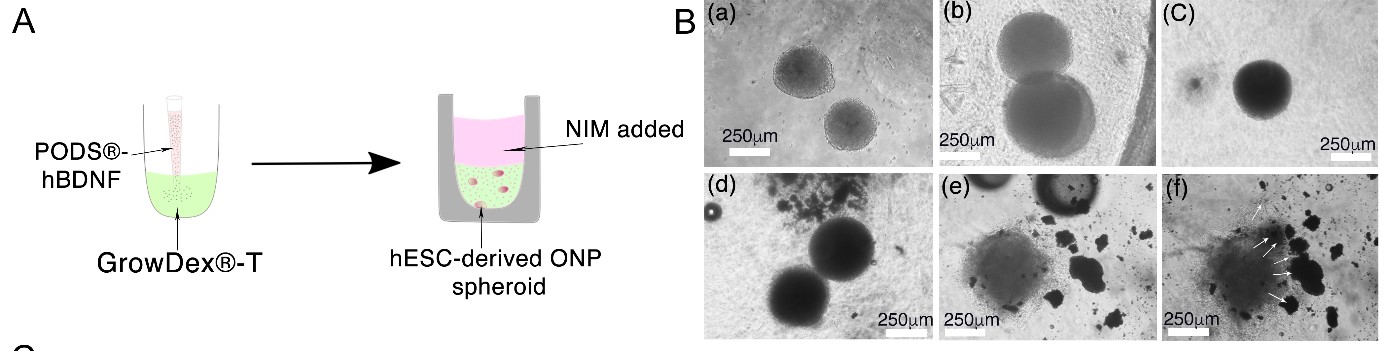
Figure 1: The schematic diagram (A) illustrates hESC-derived OPN spheroids were formed and embedded in GrowDex-T premixed with PODS-hBDNF with the addition of neuronal induction media (NIM) above the GrowDex-T. Phase contrast imaging of the OPN spheroids in GrowDex-T (B) demonstrated that they maintained their shape and integrity from 24, 48 and 72 hours without PODS-hBDNF (a-c). Whereas, when the spheroids were cultured with PODS-hBDNF (d-f), it appeared that the PODS-hBDNF may have acted as a chemo-attractant (white arrows) leading to a more differentiated phenotype.
Immunocytochemistry (ICC) staining, imaging and analysis of hESC-derived ONP spheroids was successfully performed and is illustrated in Figure 2. Spheroids were cultured in neuronal induction media (NIM) alone, GrowDex-T alone (0.5% and 0.375%), GrowDex-T (0.5% and 0.375%) with recombinant human BDNF (RhBDNF) or GrowDex-T (0.5% and 0.375%) with PODS-hBDNF.
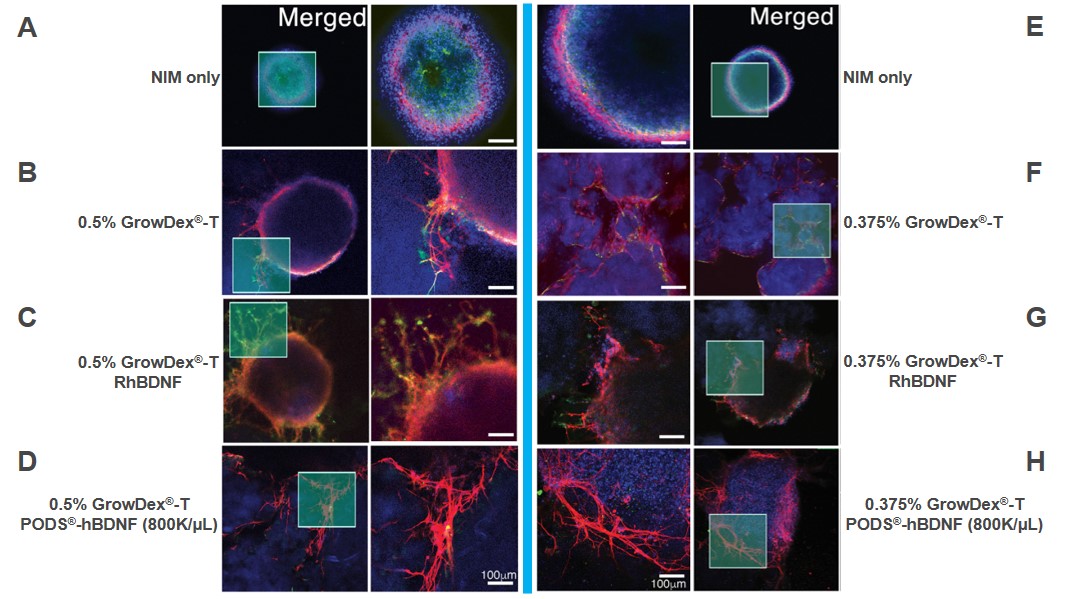
Figure 2: Immunocytochemistry of hESC-derived ONP spheroids cultured for 7 days in NIM ((A,E), GrowDex-T (0.5% B, 0.375% F), GrowDex-T with 20ng/ml RhBDNF (0.5% C, 0.375% G), or GrowDex-T with 8x105 PODS-hBDNF (0.5% D, 0.375% H). DAPI staining (blue), ICC for PAX8 (green) and β-III tubulin (red) illustrate the location and number of cells in the spheroids, differentiation capacity as well as allows for the characterization of neuronal development from the spheroids respectively).
PAX8 is an otic lineage marker. It is known to peak at the otic placode or otocyst stages of development, then downregulates after this point of differentiation. Targeted staining of PAX 8 and β-III tubulin enabled the researchers to determine the differentiation potential and neuronal network development of the hESC-derived OPNs respectively. Notably, when compared to the NIM controls, the expression of PAX8 was significantly lower in OPN spheroids cultured in a combination of GrowDex-T with either RhBDNF or PODS-hBDNF (Figure 3 A).
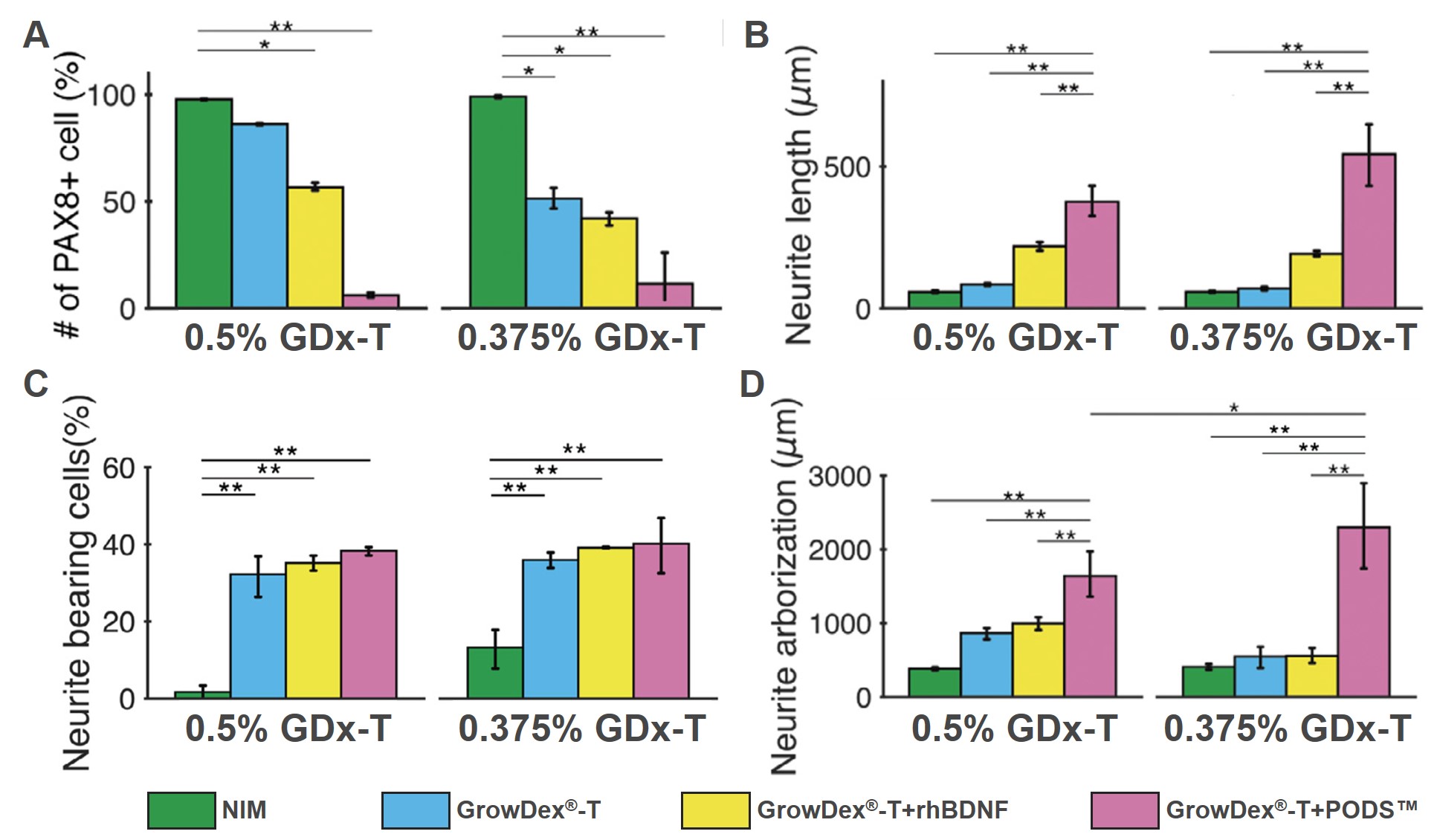
Figure 3: ICC staining of hESC-derived OPN spheroids was used for the quantification of PAX8 positivity (E), and the measurement of neurite length (F), bearing (G) and arborization analysis (H). *p < 0.05, ** p < 0.01, *** p < 0.001 by one-way ANOVA with Tukey’s post-hoc test.
Significantly longer neurites and greater neurite arborization were measured from hESC-derived ONP spheroids that were cultured in the combination of GrowDex®-T and PODS®-hBDNF (Figure 3 B, D). Additionally, ONP spheroids cultured under NIM-only conditions resulted in significantly fewer neurite-bearing cells (Figure 3 C).
CONCLUSIONS
Our protocol presented here provides sufficient evidence that we have established a robust, reproducible protocol for in vitro otic neuronal differentiation of hESC-derived ONP spheroids. Using our protocol to create an artificial stem cell niche in the mouse inner ear using 3D spheroid formation, GrowDex®-T as an ECM, and the PODS® polyhedrin delivery system for the continuous provision of BDNF, it is possible to work toward integrating transplanted hESC-derived ONPs further into the inner ear. Ultimately, this work is paving the way for the potential delivery of a regenerative medicine therapy towards repairing sensorineural hearing loss.
Results presented here have been sourced from Chang, H.-T. et al., (2020) with permission from the corresponding author.
REFERENCES
- Olusanya, B.O., et al. (2019). "Hearing loss: rising prevalence and impact." Bull World Health Organ 97(10): p. 646-646A.
- Chen, W., et al. (2012). "Restoration of auditory evoked responses by human ES-cell-derived otic progenitors." Nature 490(7419): p. 278-82.
- Matsuoka, A.J., et al. (2006). "In vivo and in vitro characterization of bone marrow-derived stem cells in the cochlea." The Laryngoscope 116(8): p. 1363-1367.
- Matsuoka, A.J., et al. (2007). "Enhanced survival of bone-marrow-derived pluripotent stem cells in an animal model of auditory neuropathy." Laryngoscope 117(9): p. 1629-35.
- Corrales, C.E., et al. (2006). "Engraftment and differentiation of embryonic stem cell-derived neural progenitor cells in the cochlear nerve trunk: growth of processes into the organ of Corti." J Neurobiol 66(13): p. 1489-500.
- Huang, E.J. and Reichardt, L.F. (2001). "Neurotrophins: roles in neuronal development and function." Annu Rev Neurosci 24: p. 677-736.
- Reichardt, L.F. (2006). "Neurotrophin-regulated signalling pathways." Philos Trans R Soc Lond B Biol Sci 361(1473): p. 1545-64.
- Matsuoka, A.J., et al. (2017). "Directed Differentiation of Human Embryonic Stem Cells Toward Placode-Derived Spiral Ganglion-Like Sensory Neurons." Stem Cells Transl Med 6(3): p. 923-936.
- Chang, H.-T., et al. (2020). "An engineered three-dimensional stem cell niche in the inner ear by applying a nanofibrillar cellulose hydrogel with a sustained-release neurotrophic factor delivery system." Acta Biomaterialia 108: p. 111-127.
Read more about Neural applications
