Photocurable Ink Formulations of GrowInks with UV Crosslinkable GelMA
Dr. Xiaoju Wang1, Prof. Chunlin Xu1, Dr. Jonathan Sheard2, Dr. Markus Nuopponen2
1Åbo Akademi University, Turku, Finland
2UPM Biomedicals, Helsinki, Finland
INTRODUCTION
Cellulose nanomaterials derived from wood biomass are emerging scaffold-candidates for biomedical hydrogels aimed towards 3D bioprinting and tissue engineering applications [1]. GrowInks are an example of nanofibrillar cellulose hydrogels which can be successfully used for 3D bioprinting [2, 3].
Methacrylated gelatine (GelMA) is an animal derived gelatine-derivative and is one of the most popular UV-curable biopolymers used in UV-aided cell-laden bioprinting. This is due to the availability of cell binding motifs, such as RGD peptides, which improve cell adhesion, migration, proliferation, and differentiation.
During and after the bioprinting process it is necessary to develop and produce 3D scaffold constructs which can maintain their structural fidelity whilst also providing the cells with an interconnected porous network for cell growth and nutrient transportation. However, inadequate rheological properties of low concentration GelMA (<5%), lead to challenges with handling and printing the hydrogel, resulting in a lack of shape and flow fidelity during the printing process [4, 5]. Therefore, GelMA is often combined with other compounds, biomaterials, or polymers to improve the handling and printability.
In this study nanofibrillar cellulose based GrowInks were mixed with GelMA and the rheological properties, photochemistry kinetics, and hydrogel material properties were characterised. Addition of GrowInk to GelMA improved the rheological properties, printability, and flow fidelity, and thus showed ease of use.
Additionally, optimized parameters and protocols were established for the hydrogels using an UV-aided direct-ink-writing (DIW) extrusion 3D printer. These optimized parameters and protocols can therefore be used to address possible application scenarios of 3D bioprinting, such as the fabrication and physiological mimicry of healthy or damaged tissue for model development or drug screening.
Some work presented in this application note is also shown in Wang, Q. et al., (2021).

MATERIALS
- GrowInk®-N (Cat No. GINK-N250-005, UPM Biomedicals)
- GrowInk®-T (Cat No. GINK-T200-005, UPM Biomedicals)
- Methacrylated gelatine (GelMA), (Cat No. GMA, ALLEVI)
- Lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP), (Cat No. 900889-1G, Sigma) used as the photo-initiator
- Phosphate-buffered saline (PBS, 1mM), (Cat No. E404-100TABS, VWR)
- ROKIT INVIVO Multi-Use Hybrid Bio 3D printer (Rokit Healthcare Inc)
- MCR 702 MultiDrive rheometer (Anton Paar GmbH) with the Anton Paar RheoCompass 1.24 software
- UV-LED 365nm (bluepoint LED eco, Hönle AG)
- Nikon ECLIPSE E200 with a Nikon DIGITAL SIGHT DS-US camera (Nikon)
- Printing Syringe: BD 3ml syringe, Luer-Lok™ Tip
- Printing Nozzle: Precision Tips (Nordson EFD), Inner diameter: 0.2 mm (27G) and 0.25 mm (25G)
- Printing Bed: Microscope slides cut edges, frosted (VWR)
- Example recommended fabrication parameters used for GrowInk-T (stock) + 5% GelMA: nozzle 25G, Input flow 110 %, print speed 12 mm/s, layer height 0.09 mm
METHODS
Ink formulations include GrowInk-N + GelMA, or GrowInk-T + GelMA. The following protocol is an example method to make 4 g of GrowInk-N + 5% GelMA + 0.1% LAP:
- Bring GelMA and LAP from cold storage, then place them along with GrowInk into a desiccator to come up to room temperature for 15 minutes.
- Formulation preparations:
- For GrowInk-N formulations, prepare 1 ml of LAP solution (10 mg/ml) by adding 10 mg LAP into 1 ml of PBS.
- For GrowInk-T-based formulations, prepare 10 mg/ml LAP using water rather than PBS. The introduction of PBS for formulation preparation of GrowInk-T + GelMA would cause deteriorated printability by clogging the nozzle, mainly due to the impact of electrostatic interactions between the charged molecules.
- Cover the container with foil and vortex for 1 min to dissolve the LAP.
- Take a clean 5 ml syringe with a luer lock.
- Add 2.4 g of GrowInk-N into the syringe.
- Add 200 mg GelMA into the syringe.
- Add 1 ml of PBS into the syringe when using GrowInk-N. Alternatively, add 1ml water when using GrowInk-T.
- Add 0.4 ml of LAP solution into the syringe.
- Soak the syringe in a water bath at 50 °C for 10 min to dissolve the GelMA and vortex for 2 min.
- Mix the GrowInk + GelMA + LAP solution by placing a needle onto the syringe and squeezing the ink into a second syringe. Then place the needle onto the second syringe and squeeze the ink back into the first syringe. Alternatively, load the hydrogel mixture into a syringe and then connect a second syringe with a luer lock, then pump the hydrogel carefully between the two syringes, trying to avoid mixing in air bubbles. Repeat the mixing step for another 3 rounds.
- Close the cap and cover the syringe with foil and store in cold storage room overnight.
- De-gas the ink. Firstly, centrifuge the syringe at 2,500 – 3,000 rpm for 10 min. Then add the ink to a 3 ml low friction syringe with luer lock. Finally, remove as much air as possible.
- Setup the UV-aided DIW extrusion printing settings as follows: set the integrated UV-LED wavelength to 365nm, at an on/off mode with 5s on and 20s off, and an intensity range of 40 ~75 mW/cm2.
- Printing parameters were as follows; nozzle diameter - 250µm, printing speed – 12mm/s, input flow – 100%.
RESULTS
The rheological properties for GrowInk with GelMA were characterized. Specifically, the shear-thinning flow behaviour (Figure 1), the viscoelastic properties (Figure 2), and the ink flow-recovery properties were measured using the thixotropic rheology method. It was noted that the addition of 5% GelMA in the form of dry powder, to both GrowInk-N and GrowInk-T, increased the viscosity (Figure 1A and 1B) and resulted in a higher yield point (Figure 1B and 1D) when compared with the pristine GrowInk. It was also observed that that the composite bioinks still maintained their shear-thinning rheological properties and printability in response to the applied shear rate, as seen by the declining trend in viscosity (ŋ) (Figure 1B and 1D).
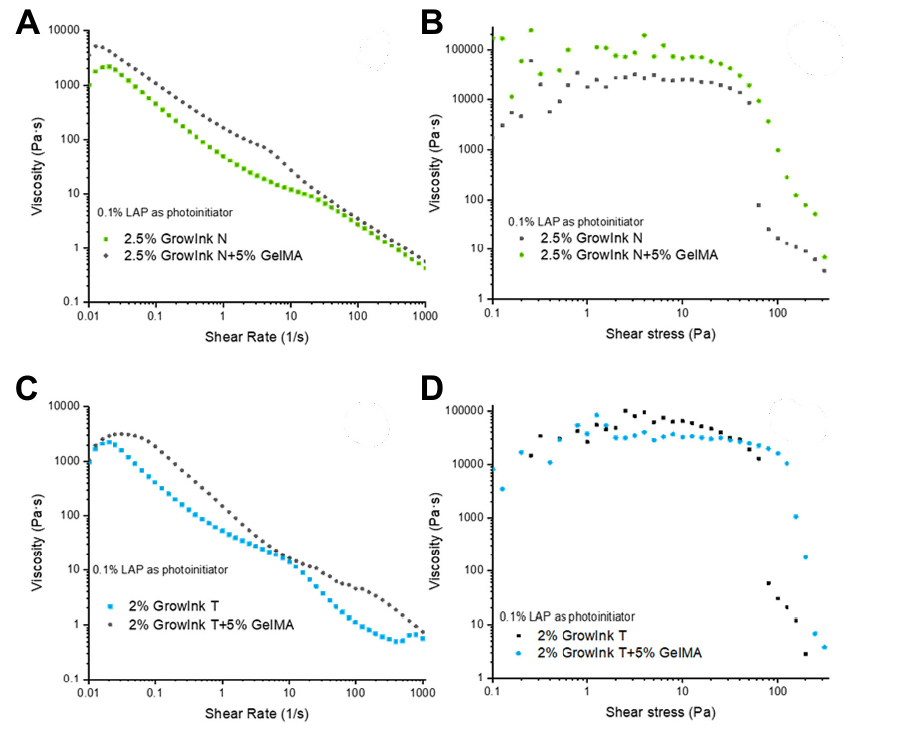
Figure 1. Shear-thinning flow curves for GrowInk-N and GrowInk-N + 5% GelMA (A), or GrowInk-T and GrowInk-T + 5% GelMA (C) measured with controlled shear rate under rotational mode. Viscosity vs shear stress curves for GrowInk-N and GrowInk-N + 5% GelMA (B), or GrowInk-T and GrowInk-T + 5% GelMA (D) measured with controlled shear stress under rotational mode.
As seen in Figure 2, the addition of 5% GelMA in the form of dry powder into GrowInks increased the G’ value (Figure 2A and 2C) and the yield stress (Figure 2B and 2D) of the formulated inks when compared with the pristine GrowInks. Especially in the case of 1.5% GrowInk-T + 5% GelMA, the yield stress increased as a result of electrostatic interaction between the negative –COO charge on cellulose nanofibers and positively charged –NH3+ in GelMA (Figure 2C and 2D).
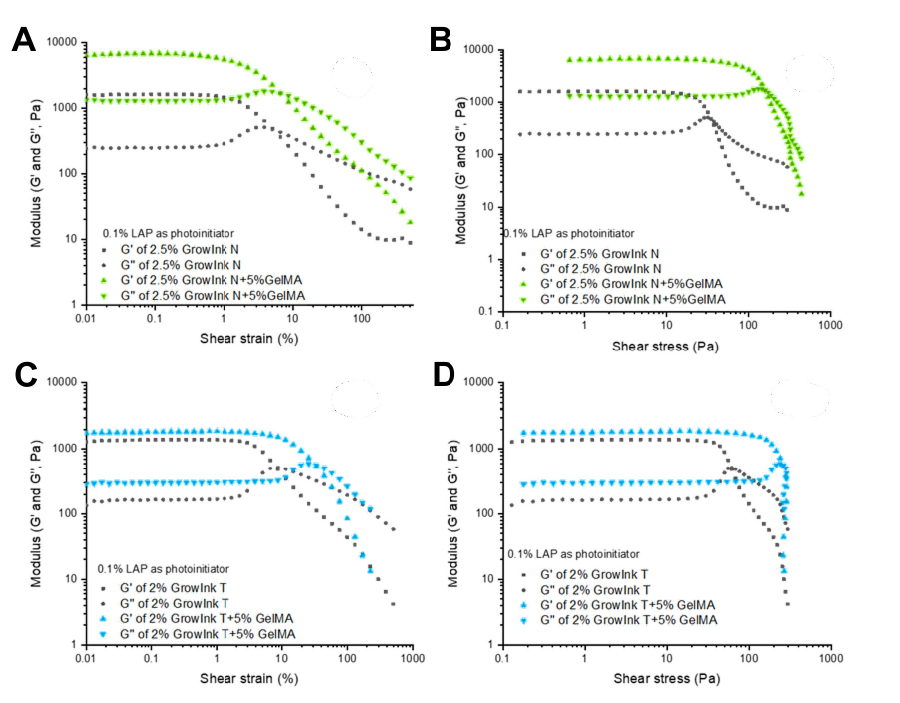
Figure 2. Viscoelastic properties, G’ and G’’ vs shear strain (A, C) and G’ and G’’ vs shear stress (B, D) measured under the oscillatory mode to acquire LVE Region for GrowInk-N and GrowInk-N + 5% GelMA (A, B), or GrowInk-T and GrowInk-T + 5% GelMA (C, D).
Printing parameters, including input flow, printing speed, and layer height, were optimized for DIW extrusion printing by assessing the ink fidelity of the respective bioink formulations using optimal microscopy, addressing both filament definition (width and uniformity) and filament diffusion. The cylindrical grid-network scaffolds shown in Figure 3 demonstrate the workability of the optimized DIW extrusion printing parameters when using GrowInk-T + 5 % GelMA as the feedstock ink material. The printing parameters were, input flow – 100%, printing speed – 12mm/s, and layer height – 0.07mm.

Figure 3. Overview of successful application: cylindrical gridnetwork hydrogel scaffold with a dimension of ф=10.5mm and z=11.2 mm printed using GrowInk-T + 5% GelMA.
CONCLUSIONS
All the GrowInk + GelMA formulations which were tested, showed shear-thinning behaviour, strong viscoelastic properties along with fast flow-recovery response after high shear. The combination of GrowInk-T with 5% GelMA supports fast UV-crosslinking, elastic interpenetrating polymeric network (IPN) and a good ink fidelity in DIW extrusion bioprinting. The properties of these bioinks support the use of these hydrogels for extrusion-based 3D bioprinting.
These optimized parameters and protocols can therefore be used to address possible application scenarios of 3D bioprinting, such as the fabrication and physiological mimicry of healthy or damaged tissue for model development, wound healing [7] or drug screening. These results demonstrate principal information for future use of GrowInks in cell-laden 3D printing scaffolds aided by photocuring.
REFERENCES
- Wang, X., et al. (2020). "Nanocellulose-Based Inks for 3D Bioprinting: Key Aspects in Research Development and Challenging Perspectives in Applications-A Mini Review." Bioengineering (Basel) 7(2).
- Noy, J., et al. "High-throughput production of GrowInk™-T models by drop-on-demand 3D bioprinting." GrowInk Application Note 1.
- Clapies, A., et al. "3D printing of GrowInk™-N and GrowInk™-T with Poietis NGB-R™ bioprinter." GrowInk Application Note 2.
- Shin, S., et al. (2017). "Cellulose Nanofibers for the Enhancement of Printability of Low Viscosity Gelatin Derivatives." 2017 12(2): p. 14.
- Xu, W., et al. (2019). "Surface Engineered Biomimetic Inks Based on UV Cross-Linkable Wood Biopolymers for 3D Printing." ACS Applied Materials & Interfaces 11(13): p. 12389-12400.
- Wang, Q., et al. (2021). "Rheological and Printability Assessments on Biomaterial Inks of Nanocellulose/Photo-Crosslinkable Biopolymer in Light-Aided 3D Printing." Frontiers in Chemical Engineering 3.
- Xu, W., et al. (2019). "On Low-Concentration Inks Formulated by Nanocellulose Assisted with Gelatin Methacrylate (GelMA) for 3D Printing toward Wound Healing Application." ACS Applied Materials & Interfaces 11(9): p. 8838-8848.