Yan-Ru Lou (Ph.D.), University of Helsinki, Finland
Introduction
Recovery of cells or spheroids for downstream processing once a live cell assay has been concluded can be advantageous for many cell based studies. Additional data relating to gene or protein expression can be generated or cells used for further experimental work such as in multi-dose studies. Here we have made use of GrowDase™ a cellulase enzyme preparation, to recover spheroids cultured in GrowDex. GrowDase is an enzyme that specifically degrades cellulose without affecting animal cells, as animal cells do not contain cellulose. The degraded products of GrowDex are nontoxic sugars. However, before using GrowDase in the 3D culture we optimized the concentration with H9-GFP, WA07 and iPS(IMR90)-4 cell lines. AlamarBlue assay was used to assess cell viability and any possible toxicity of the GrowDase enzyme. Removal of GrowDex was visualized by calcofluor white stain, which is a cellulose-binding fluorochrome. The use of H9-GFP cells enabled us to monitor the spheroid structure after the enzymatic removal of the hydrogel using confocal microscopy.
Materials
- H9-GFP modified from H9 (WA09, WiCell) WA07 and iPS(IMR90)-4 (WiCell)
- mTeSR1 medium (STEMCELL Technologies)
- GrowDex 1.5 % (UPM)
- Calcofluor white stain (Sigma FLUKA)
- GrowDase 10 mg/ml
Method
GrowDex staining and live cell imaging using a confocal microscope.
To evaluate the enzymatic removal of GrowDex, calcofluor white stain was added to the culture to stain the nanocellulose fibers following the manufacturer’s instructions. GFP fluorescence of the live H9-GFP cells and calcofluor stained GrowDex were visualized using a Leica TCS SP5II HCS A confocal microscope with Argon 488nm and UV405nm lasers, respectively.
Degradation of GrowDex hydrogel with GrowDase.
GrowDase enzyme was used to recover the 3D hPSCs and spheroids from the hydrogel which were then subsequently sub-cultured using a 2D platform following the recovery process. The optimal working temperature of the enzyme is 45–50°C, which is not suitable for human cells, therefore incubations were performed at 37°C. Before GrowDase treatment the old mTeSR1 medium was removed, and GrowDase diluted in mTeSR1 medium, and added to the samples. Various concentrations of GrowDase enzyme were evaluated during this study incubating the samples over a 24 hour period. Following enzymatic removal of GrowDex the hPSC spheroids were collected for further cultivation.
Results
The 3D spheroids stayed intact after treatment with all concentrations of the GrowDase enzyme tested. It was noted that GrowDase enzyme below 200mg/mg GrowDex was insufficient to remove the hydrogel completely.
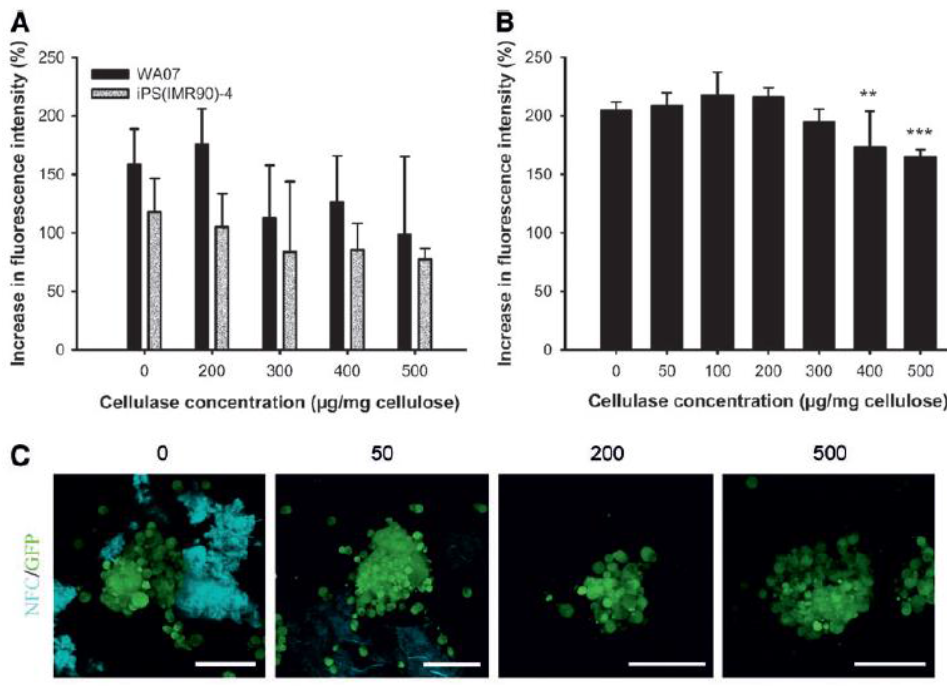
Fig. 1. Enzymatic removal of the NFC hydrogel. (A, B) Mitochondrial metabolic activity of WA07 and iPS(IMR90)-4 cells (A) and H9-GFP cells (B) as a function of the concentration of GrowDase. Cells cultured on a standard platform were treated with GrowDase at various concentrations (mg enzyme/mg hydrogel). The mitochondrial metabolic activity was determined by an AlamarBlue assay, and the increase in fluorescence intensity after a 24-h treatment with GrowDase is presented. Four (A) or six (B) biological samples for each condition were prepared. The results are expressed as mean ± standard deviation (SD). (C) Removal of GrowDex by 0, 50, 200, and 500 µg enzyme/mg hydrogel is visualized by calcofluor white staining of GrowDex, which is represented in blue. H9-GFP cells are seen in green. Adapted with permission from (1). Copyright 2014, Mary Ann Liebert, Inc.
Conclusions
In this study we have demonstrated that GrowDex can be removed from cell samples by enzymatic digestion using the GrowDase enzyme. Here spheroids were successfully recovered from the 3D samples for further downstream processing or re-culture. In addition, no significant impact on cell viability was observed as a result of performing the recovery process, making the use of GrowDex in combination with GrowDase as ideal system for 3D culture and cell recovery.
Reference
(1) Lou Y-R et al. 2014 "The Use of Nanofibrillar Cellulose Hydrogel As a Flexible Three-Dimensional Model to Culture Human Pluripotent Stem Cells. STEM CELLS AND DEVELOPMENT 23:4, pp.380-392.