Viktoria Zaderer1, Martin Hermann2, Cornelia Lass-Flörl1, Wilfried Posch1 and Doris Wilflingseder1
1Institute of Hygiene and Medical Microbiology, Medical University of Innsbruck, Austria 2Department of Anesthesiology and Critical Care Medicine, Medical University of Innsbruck, Austria
INTRODUCTION
Advanced in vitro cell models with polarized growth of respiratory epithelial cells are required for respiratory disease research, drug screening or host pathogen interaction studies. Three-dimensional (3D) cell culture in hydrogels offers a promising platform for the development of these models, however, there are challenges with the materials and methods currently used. Namely, seeding the cells in collagen-based matrices on to the inside membrane of a Transwell insert makes it practically impossible for imaging. Additionally, harvesting cells with collagenase or dispase can seriously affect the cells, resulting in biased downstream analyses [1].
To overcome these challenges, we developed and optimized long-term culturing conditions for monitoring cell differentiation and repeated dose drug response in an advanced 3D respiratory cell model [2]. Specifically, Normal Human Bronchial Epithelial (NHBE) or Small Airway Epithelial (SAE) cells were seeded upside-down on to the underside membrane of a Transwell inserts within the animal- free nanofibrillar cellulose hydrogel, GrowDex®. The cells were grown inverted under static conditions and subsequently differentiated in an air-liquid interphase (ALI) over a period of 14 days and then maintained for up to 700 days, allowing this ‘upside-down’ cell culture model to be easily monitored by live cell imaging. This method of model orientation also enables the easy addition of immune components, such as dendritic cells (DCs), macrophages, and neutrophil to the inner chamber of the Transwell inserts, to monitor immune cell behaviour after respiratory challenge.
MATERIALS
- GrowDex® 1.5% (UPM Biomedicals, Finland, Cat No. 100 103 005)
- Human derived respiratory epithelial primary cells:
- NHBE: Cat CC-2540-S (Lonza, Germany)
- SAEC: Cat CC-2547-S (Lonza, Germany)
- Complete PneumaCult™-Ex Plus Medium (STEMCELL, Germany, Cat 05041 with 05042)
- Complete PneumaCult™-ALI Base Medium (STEMCELL, Germany, Cat 05002 with 05003)
- Complete PneumaCult™-ALI Maintenance Medium (STEMCELL, Germany, Cat No. 05002 with 05006)
- Supplemented with Hydrocortisone stock solution (STEMCELL, Germany, Cat 07925) and Heparin Solution (STEMCELL, Germany, Cat No. 07980)
- Animal Component-free cell dissociation kit (STEMCELL, Germany, Cat 05426)
- WGA 488 (Biotium, USA, Cat 29022-1)
- Hoechst 33342 (Cell signaling Technology, Germany, Cat 4082)
- Phalloidin-Alexa555 (Cell Signaling Technology, Germany, Cat 8953)
- Mitotracker deep red (Life Technologies, Austria, Cat M22426)
- Costar® 6 and 24-well Clear TC-treated Multiple Well Plates (Corning Costar, The Netherlands, Cat 3526)
- 5 mm Transwell® with 8.0 µm Pore Polycarbonate Membrane Insert for 24 well plates (Corning Costar, The Netherlands, Cat No. 3422)
- Operetta CLS™ HCS system (Perkin Elmer Cellular Technologies, Germany)
- Confocal microscope (LEICA SP5, Germany)
METHODS
- GrowDex Solution for membrane coating
- Pre-warm Complete PneumaCultTM Ex Plus Medium in the water bath at 37°C. To seed NHBE or SAE cells upside-down in the Transwell inserts, a 0.5% GrowDex solution is used. Thus, for preparing 1 mL of 0.5% GrowDex, 333 µL of 1.5% stock solution is gently mixed with 567 µl of pre-warmed cell culture medium and 100 µL of cell suspension (1x107 cells/ml).
- Mix components gently by pipetting up and down and try to avoid air
- Upside-down seeding of NHBE or SAE cells in GrowDex
- 1x105 cells are seeded per Transwell insert in 100 µL of 0.5% GrowDex.
- Transwell inserts are secured in 6-well plates upside-down by taping the overlaying edge to the bottom of the Do not touch the membranes facing upwards.
- Add 100 µL of 0.5% GrowDex/cell mixture to the membranes (to the underside which is facing upwards).
- For the overnight incubation of upside-down seeded cells, cover cells with medium from the apical Avoid formation of air bubbles.
- Then flip the Transwell insert with cells in GrowDex to the normal orientation and place the insert into a single well of a 24-well plate and culture the cells for 3 days under submerged
- Remove medium from the lower compartment to produce Air-Liquid interface (ALI) and add 250 µL ALI Maintenance Medium to the top of the membrane in the center of the tissue culture insert.
- Replace medium every other day.
- Live cell imaging
- For monitoring the viability and differentiation of the cells, perform live cell imaging by preparing a master mix with Hoechst 33342 (nuclei), WGA-488 (surface structures/cilia), and Mitotracker diluted in D-PBS.
- Hoechst 33342 2 µg/mL
- WGA-488 5 µg/mL
- Mitotracker in far red 100–500 nM
- Add 50-100 µL of the master mix to the bottom of a glass-bottom
- Transfer a Transwell insert with GrowDex culture directly to the master mix
- Imaging can be started immediately, but fluorescence intensities increase with
- Transwell inserts can be transferred to imaging plates without cutting the membrane and after microscopy they can be taken back into the culture without any harm to the
- For monitoring the viability and differentiation of the cells, perform live cell imaging by preparing a master mix with Hoechst 33342 (nuclei), WGA-488 (surface structures/cilia), and Mitotracker diluted in D-PBS.
METHODS
Upside-down seeding of primary respiratory cells within GrowDex on an inverted Transwell inserts provides an efficient method for monitoring cellular proliferation and differentiation over a long period of time (Fig. 1). The method enables the long-term repeated analysis of the same cells under live conditions and for multiple drug response exposures.
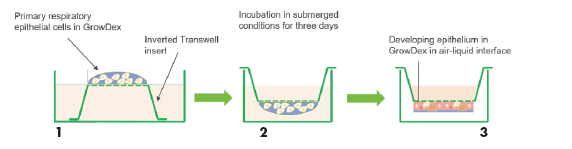
Figure 1. Schematic presentation of seeding primary respiratory cells in GrowDex for upside-down culture on Transwell inserts. Cells are mixed in GrowDex and transferred to the underside of an inverted Transwell insert which is secured in a 6-well plate (1). Cells are incubated at 37°C overnight and on the next day the insert with cells is flipped to normal orientation and placed into a well in a 24-well plate (2). Cells are cultured submerged in culture medium for 3 days and then differentiation is continued by exposing them to an air-liquid interface (ALI) by removing the medium from the bottom chamber (3).
The upside-down cultures of the NHBE respiratory epithelial cells in GrowDex showed considerably faster proliferation by quantification of the total number of nuclei (Fig. 2A), and the clear presence of differentiation cells (Fig. 2B) compared to the same cells cultured with rat-tail collagen. It was also noted that the model was sufficiently mature for further experiments after only two weeks in culture. NHBE cells also showed high levels of mature cilia with mucociliary clearance and mucus production (data not shown, please refer to the full article Zaderer, V. et al., (2019) [2]).
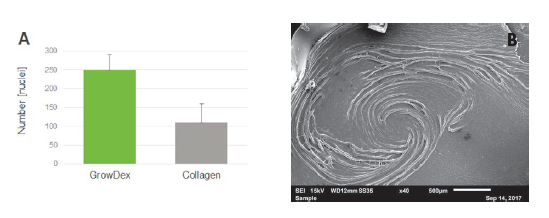
Figure 2. A) Quantitative analysis of cell nuclei in GrowDex and rat-tail collagen on day 15 post-ALI. Differentiated cells revealed a significantly higher cell number in GrowDex. Analysis was performed on images taken from 5 independent regions of 2 Transwell inserts using Operetta CLS imaging system. B) SEM image of differentiating NHBE cells in GrowDex.
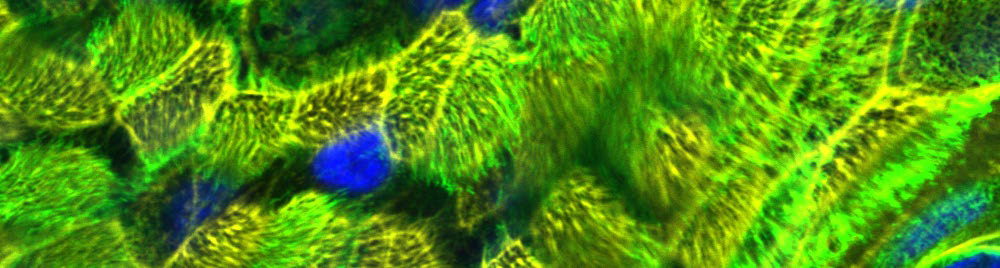
Intact ciliated pseudostratified epithelia were successfully cultured for over 700 days, thereby making this 3D respiratory model best fitted for long-term repeated exposure experiments to monitor novel drugs or compounds over a 2-year period (Fig. 3).
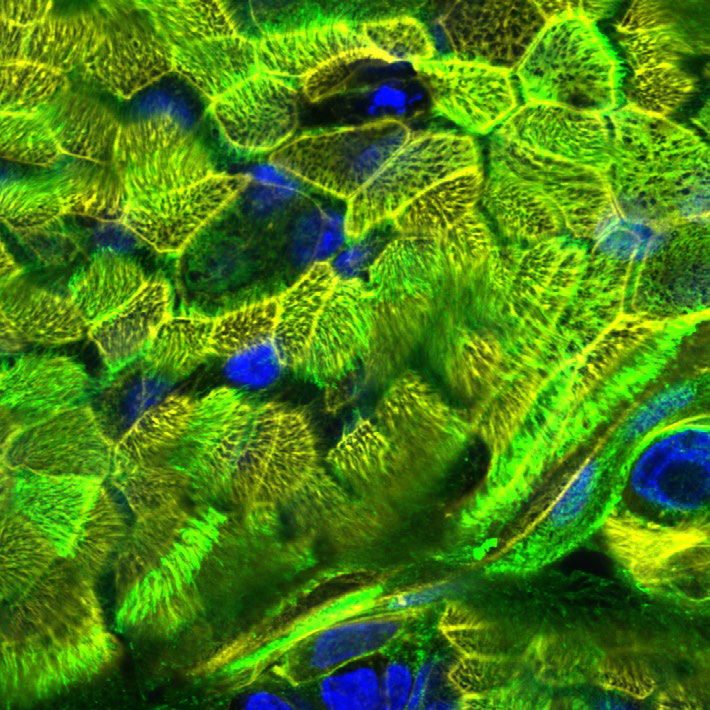
Figure 3. Epithelial integrity of upside-down cultured NHBE cells in GrowDex after 700 d culture. Cilia of the cells were stained using WGA-488 (green), cytoskeleton using Phalloidin-Alexa555 (yellow), and nuclei using Höchst (blue).
CONCLUSIONS
The protocol and data presented here shows the successful development of an advanced animal-free and optimized long-term 3D respiratory cell model, where NHBE cells are cultured towards a polarized, fully differentiated epithelium, whilst culture conditions allow for monitoring cell differentiation and repeated dose drug responses. Such cultured 3D models offer an excellent platform for host-pathogen interaction or drug efficacy studies. Without animal-derived materials in the system, unspecific or cross- species reactivity, and matrix batch related variations can be avoided. Furthermore, the system allows repeated analysis of the same cultures as the complete Transwell insert can be transferred for live cell imaging, and then returned to its original plate for further cultivation. Additionally, it is possible to simply add any desired immune cells into the upper chamber of the insert whilst applying a stimulus directly into the lower chamber or glass-bottom dish to monitor real-time immune cell behavior upon activation. Also, for more detailed downstream analysis of the cells e.g. with flow cytometry, it is important to be able harvest the cells from the culture matrix. GrowDex hydrogel can be easily digested by using cellulase enzyme - GrowDase, without affecting the cells.
In summary, culturing human derived respiratory epithelial primary cells in GrowDex hydrogel in the presented upside-down setup, not only enables the extended culture of the cells for over 700 days, but also facilitates live cell analyses of the same cells in repeated dose experiments with drug compounds, whilst allowing for the development of the model into a more complex system with the addition of relevant immune cells and subsequent experimental readouts. This system forms a robust platform for respiratory disease research, host-pathogen interaction studies, as well as for drug efficacy screens.
REFERENCES
- Autengruber, A., et al. (2012). "Impact of enzymatic tissue disintegration on the level of surface molecule expression and immune cell " European journal of microbiology & immunology 2(2): p. 112-120.
- Zaderer, , et al. (2019). "Turning the World Upside-Down in Cellulose for Improved Culturing and Imaging of Respiratory Challenges within a Human 3D Model." Cells 8(10): p. 1292.