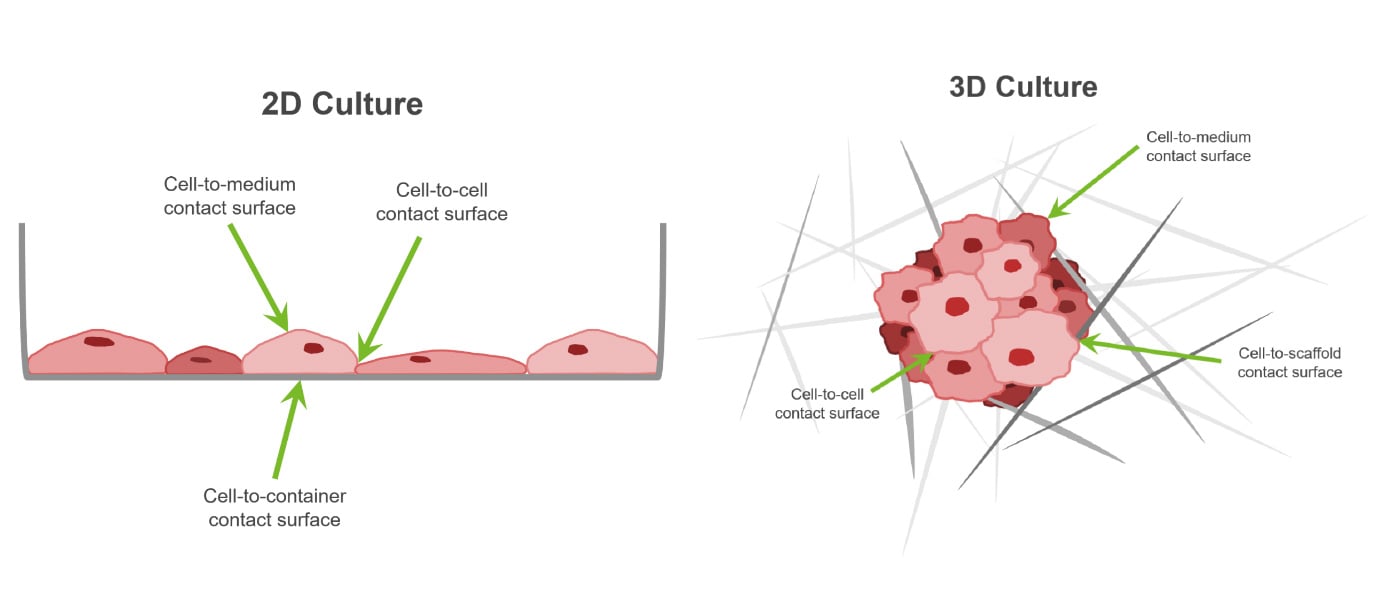
For mammalian cells to be studied, they need to be grown in an environment that mimics the body as closely as possible. This has traditionally been done by growing the cells on a flat surface, in other words, in two dimensions. This has many limitations, namely that the cells inside the body do not grow in 2D, but in 3D, which has surprisingly big implications on how the cells behave.
You might be familiar with the rapidly growing popularity of 3D cell culture, but you still have some questions left unanswered. On this page you will find what the main differences are between 2D and 3D, their advantages and disadvantages, why you might want to choose 3D, which 3D culture format might best suit your needs, and finally, how to get started on 3D cell culture.